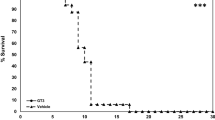
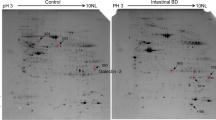
Abstract
Background and Aims
Radiation enteritis (RE) has emerged as a significant complication that can progress to severe gastrointestinal disease and the mechanisms underlying its genesis remain poorly understood. The aim of this study was to identify temporal changes in protein expression potentially associated with acute inflammation and to elucidate the mechanism underlying radiation enteritis genesis.
Methods
Male Sprague–Dawley rats were irradiated in the abdomen with a single dose of 10 Gy to establish an in vivo model of acute radiation enteritis. Two-dimensional fluorescence difference gel electrophoresis, matrix-assisted laser desorption/ionization time-of-flight spectrometer (MALDI-TOF) tandem mass spectrometry, and peptide mass fingerprinting were used to determine differentially expressed proteins between normal and inflamed intestinal mucosa. Additionally, differentially expressed proteins were evaluated by KO Based Annotation System to find the biological functions associated with acute radiation enteritis.
Results
Intensity changes of 86 spots were detected with statistical significance (ratio ≥ 1.5 or ≤ 1.5, P < 0.05). Sixty one of the 86 spots were identified by MALDI-TOF/TOF tandem mass spectrometry. These radiation-induced proteins with biological functions showed that the FAS pathway and glycolysis signaling pathways were significantly altered using the KOBAS tool.
Conclusions
Our results reveal an underlying mechanism of radiation-induced acute enteritis, which may help clarify the pathogenesis of RE and point to potential targets for therapeutic interventions.




Similar content being viewed by others
References
Buchi K. Radiation proctitis: therapy and prognosis. JAMA. 1991;265:1180.
Ooi BS, Tjandra JJ, Green MD. Morbidities of adjuvant chemotherapy and radiotherapy for resectable rectal cancer: an overview. Dis Colon Rectum. 1999;42:403–418.
McGough C, Baldwin C, Frost G, Andreyev HJ. Role of nutritional intervention in patients treated with radiotherapy for pelvic malignancy. Br J Cancer. 2004;90:2278–2287.
Andreyev HJ. Gastrointestinal problems after pelvic radiotherapy: the past the present and the future. Clin Oncol R Coll Radiol. 2007;19:790–799.
Ghezzi F, Cromi A, Serati M, et al. Radiation-induced bowel complications: laparoscopic versus open staging of gynecologic malignancy. Ann Surg Oncol. 2011;18:782–791.
Eifel PJ, Levenback C, Wharton JT, Oswald MJ. Time course and incidence of late complications in patients treated with radiation therapy for FIGO stage IB carcinoma of the uterine cervix. Int J Radiat Oncol Biol Phys. 1995;32:1289–1300.
Potten CS, Grant HK. The relationship between ionizing radiation-induced apoptosis and stem cells in the small and large intestine. Br J Cancer. 1998;78:993–1003.
Nguyen NP, Antoine JE, Dutta S, Karlsson U, Sallah S. Current concepts in radiation enteritis and implications for future clinical trials. Cancer. 2002;95:1151–1163.
Anderson NL, Anderson NG. Proteome and proteomics: new technologies, new concepts, and new words. Electrophoresis. 1998;19:1853–1861.
Chambers G, Lawrie L, Cash P, Murray GI. Proteomics: a new approach to the study of disease. J Pathol. 2000;192:280–288.
Hanash S. Disease proteomics. Nature. 2003;422:226–232.
Zhou G, Li H, DeCamp D, et al. 2D differential in-gel electrophoresis for the identification of esophageal scans cell cancer-specific protein markers. Mol Cell Proteomics. 2002;1:117–124.
Roxo-Rosa M, da Costa G, Luider TM, et al. Proteomic analysis of nasal cells from cystic fibrosis patients and non-cystic fibrosis control individuals: search for novel biomarkers of cystic fibrosis lung disease. Proteomics. 2006;6:2314–2325.
Zhang X, Yang J, Guo Y, et al. Functional proteomic analysis of nonalcoholic fatty liver disease in rat models: enoyl-coenzyme a hydratase down-regulation exacerbates hepatic steatosis. Hepatology. 2010;51:1190–1199.
Klimberg VS, Salloum RM, Kasper M, et al. Oral glutamine accelerates healing of the small intestine and improves outcome after whole abdominal radiation. Arch Surg. 1990;125:1040–1045.
Howarth GS, Fraser R, Frisby CL, Schirmer MB, Yeoh EK. Effects of insulin-like growth factor-I administration on radiation enteritis in rats. Scand J Gastroenterol. 1997;32:1118–1124.
Luo YX, Cui J, Wang L, et al. Identification of cancer-associated proteins by proteomics and downregulation of beta-tropomyosin expression in colorectal adenoma and cancer. Proteomics Clin Appl. 2009;3:1397–1406.
Jin S, Shen JN, Guo QC, et al. 2-D DIGE and MALDI-TOF-MS analysis of the serum proteome in human osteosarcoma. Proteomics Clin Appl. 2007;1:272–285.
Xie C, Mao X, Huang J, et al. KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 2011;39:W316–W322.
Wu J, Mao X, Cai T, Luo J, Wei L. KOBAS server: a web-based platform for automated annotation and pathway identification. Nucleic Acids Res. 2006;34:W720–W724.
Guicciardi ME, Gores GJ. Apoptosis: a mechanism of acute and chronic liver injury. Gut. 2005;54:1024–1033.
Kazama H, Yonehara S. The mechanism and regulation of Fas-mediated apoptosis. Seikagaku. 2001;73:107–111.
Shimada K, Nakamura M, Ishida E, Kishi M, Konishi N. Androgen and the blocking of radiation-induced sensitization to Fas-mediated apoptosis through c-jun induction in prostate cancer cells. Int J Radiat Biol. 2003;79:451–462.
DosReis GA, Borges VM. Role of Fas-ligand induced apoptosis in pulmonary inflammation and injury. Curr Drug Targets Inflamm Allergy. 2003;2:161–167.
Uno M, Otsuki T, Yata K, et al. Participation of Fas-mediated apoptotic pathway in KB, a human head and neck squamous cell carcinoma cell line, after irradiation. Int J Oncol. 2002;20:617–622.
Takahashi H, Ishida-Yamamoto A, Iizuka H. Ultraviolet B irradiation induces apoptosis of keratinocytes by direct activation of Fas antigen. J Investig Dermatol Symp Proc. 2001;6:64–68.
Yang JY, Xia W, Hu MC. Ionizing radiation activates expression of FOXO3a, Fas ligand, and Bim, and induces cell apoptosis. Int J Oncol. 2006;29:643–648.
Reztsova VV. Role of glycolysis in initiation of immortality and apoptosis. Vopr Onkol. 2006;52:609–615.
Sukhomlinov BF, Chaika Ia P, Monastyrskaia SS, Demida EN. The individual glycolysis enzymes in the enterocytes of the rat small intestine studied during exposure to ionizing radiation. Radiobiologiia. 1993;33:255–258.
Ralser M, Wamelink MM, Kowald A, et al. Dynamic rerouting of the carbohydrate flux is key to counteracting oxidative stress. J Biol. 2007;6:10.
Grant CM. Metabolic reconfiguration is a regulated response to oxidative stress. J Biol. 2008;7:1.
Otsuki S, Morshed SR, Chowdhury SA, et al. Possible link between glycolysis and apoptosis induced by sodium fluoride. J Dent Res. 2005;84:919–923.
Robinson GL, Dinsdale D, Macfarlane M, Cain K. Switching from aerobic glycolysis to oxidative phosphorylation modulates the sensitivity of mantle cell lymphoma cells to TRAIL. Oncogene. 2012;31:4996–5006.
Zhong J, Rajaram N, Brazil DM, et al. Radiation induces aerobic glycolysis through reactive oxygen species. Radiother Oncology. 2013;1:390–396.
Wajant H. The Fas signaling pathway: more than a paradigm. Science. 2002;296:1635–1636.
Brint E, O’Callaghan G, Houston A. Life in the Fas lane: differential outcomes of Fas signaling. Cell Mol Life Sci. 2013;70:4085–4099.
Fais S, De Milito A, Lozupone F. The role of FAS to ezrin association in FAS-mediated apoptosis. Apoptosis. 2005;10:941–947.
Kuwabara M, Takahashi K, Inanami O. Induction of apoptosis through the activation of SAPK/JNK followed by the expression of death receptor Fas in X-irradiated cells. J Radiat Res. 2003;44:203–209.
Dzietko M, Boos V, Sifringer M, et al. A critical role for Fas/CD-95 dependent signaling pathways in the pathogenesis of hyperoxia-induced brain injury. Ann Neurol. 2008;64:664–673.
Zhuang S, Kochevar IE. Ultraviolet A radiation induces rapid apoptosis of human leukemia cells by Fas ligand-independent activation of the Fas death pathways. Photochem Photobiol. 2003;78:61–67.
Reap EA, Roof K, Maynor K, Borrero M, Booker J, Cohen PL. Radiation and stress-induced apoptosis: a role for Fas/Fas ligand interactions. Proc Natl Acad Sci USA. 1997;94:5750–5755.
Ao X, Lubman DM, Davis MA, et al. Comparative proteomic analysis of radiation-induced changes in mouse lung: fibrosis-sensitive and -resistant strains. Radiat Res. 2008;169:417–425.
Acknowledgments
This study was partly supported by the Guangdong Provincial Key Scientific Research Grants (10251008901000008, L Wang), the National Natural Scientific Foundation of China Grants (81072042, L Wang; 81172040, JP Wang), and grants by ”985 project” of Sun Yat-Sen University and Guangdong Translational Medicine Public Platform (4202037) and Sun Yat-Sen Graduate Innovative Personnel Training Funded Projects (Shunxin Song and Jianping Wang). We thank Ms. Biyan Lu, Peihuang Wu, Dr. Kunhua Hu and Chuangyu Wen for their laboratory assistance.
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Song, S., Chen, D., Ma, T. et al. Molecular Mechanism of Acute Radiation Enteritis Revealed Using Proteomics and Biological Signaling Network Analysis in Rats. Dig Dis Sci 59, 2704–2713 (2014). https://doi.org/10.1007/s10620-014-3224-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3224-1