Abstract
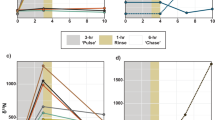
Bioconcentration kinetics of five metals (Ag, Cd, Co, Mn, and Zn) were determined in the scleractinian coral Stylophora pistillata (entire symbiotic association vs. cultured symbionts), using radiotracer techniques. Among contrasting element behaviors observed in S. pistillata, the highest efficiency of concentration and retention was observed for Ag in the symbiotic association (CFss reaching 5000 and T b½ > 1 year). Predominant proportion of this metal was found associated with the skeleton whereas the other metals were mainly present in the coral tissues (including host tissues and symbionts). A 96-h exposure of cultured symbionts (isolated zooxantellae from S. pistillata) indicated that they displayed a very high potential for metal bioconcentration (higher by 1 to 3 orders of magnitude compared to the skeleton). In addition, among the five elements investigated, Ag had the highest concentration factor in the cultured symbionts. Contrasting kinetic characteristics of skeleton vs. tissues offer interesting implications for biomonitoring purposes. Indeed, the skeleton was shown to display stable metal concentrations after an exposure (long retention time) and thereby allows recording contamination event on the long term, whereas the concentrations within coral tissues rapidly increased during the exposure and dropped when non-contaminating conditions were restored, allowing information on the current (short term) contamination status. The present study confirms that the coral can be seen as a two-compartment box model for metal bioconcentration: the tissues sensus latto as a first box governing metal entrance (with a crucial role played by the symbionts) and the skeleton as a second box where metal detoxification (storage) is taking place; the first box also depurates toward the environment when non-contaminating conditions are restored.




Similar content being viewed by others
References
Ateweberhan, M., Feary, D. A., Keshavmurthy, S., Chen, A., Schleyer, M. H., & Sheppard, C. R. C. (2013). Climate change impacts on coral reefs: synergies with local effects, possibilities for acclimation, and management implications. Marine Pollution Bulletin, 74(2), 526–539. doi:10.1016/j.marpolbul.2013.06.011.
Baker, A. C. (2004). Corals’ adaptive response to climate change. Nature, 430, 741.
Bielmyer, G. K., Grosell, M., Bhagooli, R., Baker, A. C., Langdon, C., Gillette, P., & Capo, T. R. (2010). Differential effects of copper on three species of scleractinian corals and their algal symbionts (Symbiodinium spp.). Aquatic Toxicology (Amsterdam, Netherlands), 97(2), 125–133. doi:10.1016/j.aquatox.2009.12.021.
Brown, B. E., Tudhope, A. W., Tissier, M. D. A., & Scoffin, T. P. (1991). A novel mechanism for iron incorporation into coral skeletons. Coral Reefs, 10(4), 211–215.
Esslemont, G., Harriott, V. J., & McConchie, D. M. (2000). Variability of trace-metal concentrations within and between colonies of Pocillopora damicornis. Marine Pollution Bulletin, 40(7), 637–642. doi:10.1016/S0025-326X(00)00068-0.
Ferrier-Pagès, C., Houlbrèque, F., Wyse, E., Richard, C., Allemand, D., & Boisson, F. (2005). Bioaccumulation of zinc in the scleractinian coral Stylophora pistillata. Coral Reefs, 24(4), 636–645. doi:10.1007/s00338-005-0045-x.
Fisher, N. S. (1985). Accumulation of metals by marine picoplankton. Marine Biology, 87(2), 137–142.
Hanna, R. G., & Muir, G. L. (1990). Red Sea corals as biomonitors of trace metal pollution. Environmental Monitoring and Assessment, 14, 211–222.
Hédouin, L., & Gates, R. D. (2013). Assessing fertilization success of the coral Montipora capitata under copper exposure: does the night of spawning matter? Marine Pollution Bulletin, 66(1–2), 221–224.
Hédouin, L., Gomez Batista, M., Metian, M., Buschiazzo, E., & Warnau, M. (2010). Metal and metalloid bioconcentration capacity of two tropical bivalves for monitoring the impact of land-based mining activities in the New Caledonia lagoon. Marine Pollution Bulletin, 61(7–12), 554–567.
Houlbrèque, F., & Ferrier-Pagès, C. (2009). Heterotrophy in tropical scleractinian corals. Biological Reviews of the Cambridge Philosophical Society, 84(1), 1–17. doi:10.1111/j.1469-185X.2008.00058.x.
Houlbrèque, F., Rodolfo-Metalpa, R., Jeffree, R., Oberhänsli, F., Teyssié, J.-L., Boisson, F., et al. (2011). Effects of increased pCO2 on zinc uptake and calcification in the tropical coral Stylophora pistillata. Coral Reefs, 31(1), 101–109. doi:10.1007/s00338-011-0819-2.
Hughes, T. P., Baird, A. H., Bellwood, D. R., Card, M., Connolly, S. R., Folke, C., et al. (2003). Climate change, human impacts, and the resilience of coral reefs. Science, 301, 929–933. doi:10.1126/science.1085046.
Lacoue-Labarthe, T., Warnau, M., Metian, M., Oberhänsli, F., Rouleau, C., & Bustamante, P. (2009). Biokinetics of Hg and Pb accumulation in the encapsulated egg of the common cuttlefish Sepia officinalis: radiotracer experiments. Science of the Total Environment, 407(24), 6188–6195.
Livingston, H. G., & Thompson, G. (1971). Trace element concentrations in some modern corals. Limnology and Oceanography, 16, 786–796.
Marshall, A. (2002). Occurrence, distribution, and localisation of metals in cnidarians. Microscopy Research and Technique, 56, 341–357.
Metian, M., Bustamante, P., Cosson, R. P., Hédouin, L., & Warnau, M. (2008). Investigation of Ag in the king scallop Pecten maximus using field and laboratory approaches. Journal of Experimental Marine Biology and Ecology, 367(1), 53–60.
Metian, M., & Warnau, M. (2008). The tropical brown alga Lobophora variegata: a prospective bioindicator for Ag contamination in Tropical coastal waters. Bulletin of Environmental Contamination and Toxicology, 81(5), 455–458.
Mitchelmore, C. L., Verde, E. A., & Weis, V. M. (2007). Uptake and partitioning of copper and cadmium in the coral Pocillopora damicornis. Aquatic Toxicology, 85(1), 48–56. doi:10.1016/j.aquatox.2007.07.015.
Moberg, F., & Folke, C. (1999). Ecological goods and services of coral reef ecosystems. Ecological Economics, 29, 215–233. doi:10.1016/S0921-8009(99)00009-9.
Nystrom, M., Nordemar, I., & Tedengren, M. (2001). Simultaneous and sequential stress from increased temperature and copper on the metabolism of the hermatypic coral Porites cylindrica. Marine Biology, 138, 1225–1231.
Peters, E. C., Gassman, N. J., Firman, J. C., Richmond, R. H., & Power, E. A. (1997). Ecotoxicology of tropical marine ecosystems. Environmental Toxicology and Chemistry, 16(1), 12–40.
Ratte, H. T. (1999). Bioaccumulation and toxicity of silver compounds, a review. Environmental Toxicology and Chemistry, 18, 98–108.
Redding, J. E., Myers-Miller, R. L., Baker, D. M., Fogel, M., Raymundo, L. J., & Kim, K. (2013). Link between sewage-derived nitrogen pollution and coral disease severity in Guam. Marine Pollution Bulletin, 73(1), 57–63. doi:10.1016/j.marpolbul.2013.06.002.
Reichelt-Brushett, A. J., & Harrison, P. L. (2000). The effect of copper on the settlement success of larvae from the scleractinian coral Acropora tenuis. Marine Pollution Bulletin, 41, 385–391.
Reichelt-Brushett, A. J., & McOrist, G. (2003). Trace metals in the living and nonliving components of scleractinian corals. Marine Pollution Bulletin, 46(12), 1573–1582. doi:10.1016/S0025-326X(03)00323-0.
Rogers, C. S. (1990). Responses of coral reefs and reef organisms to sedimentation. Marine Ecology Progress Series, 62, 185–202.
Sañudo-Wilhelmy, S. A., & Flegal, R. (1992). Anthropogenic silver in the southern California bight: a new tracer of sewage in coastal waters. Environmental Science and Technology, 26, 2147–2151.
Shen, G. T., & Boyle, E. A. (1988). Determination of lead, cadmium and other trace metals in annually-banded corals. Chemical Geology, 67(1–2), 47–62. doi:10.1016/0009-2541(88)90005-8.
Tambutté, E., Allemand, D., Bourge, I., Gattuso, J.-P., & Jaubert, J. (1995). An improved 45Ca protocol for investigating physiological mechanisms in coral calcification. Marine Biology, 122, 453–459.
Ward, S., & Harrison, P. (2000). Changes in gametogenesis and fecundity of acroporid corals that were exposed to elevated nitrogen and phosphorus during the ENCORE experiment. Journal of Experimental Marine Biology and Ecology, 246(2), 179–221.
Zar, J. H. (1996). Biostatistical analysis. Upper Saddle River: Prentice Hall.
Acknowledgments
The IAEA is grateful for the support provided to its Environment Laboratories by the Government of the Principality of Monaco. MW is an Honorary Senior Research Associate of the National Fund for Scientific Research (NFSR, Belgium). LH was beneficiary of a PhD grant (CIFRE, France) supported by the Goro-Nickel Company, New Caledonia. This project was also funded by the Scientific Center of Monaco (and Monaco government).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Metian, M., Hédouin, L., Ferrier-Pagès, C. et al. Metal bioconcentration in the scleractinian coral Stylophora pistillata: investigating the role of different components of the holobiont using radiotracers. Environ Monit Assess 187, 178 (2015). https://doi.org/10.1007/s10661-015-4383-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4383-z