Abstract
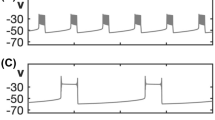
Gonadotropin-releasing hormone (GnRH) neurons exhibit at least two intrinsic modes of action potential burst firing, referred to as parabolic and irregular bursting. Parabolic bursting is characterized by a slow wave in membrane potential that can underlie periodic clusters of action potentials with increased interspike interval at the beginning and at the end of each cluster. Irregular bursting is characterized by clusters of action potentials that are separated by varying durations of interburst intervals and a relatively stable baseline potential. Based on recent studies of isolated ionic currents, a stochastic Hodgkin-Huxley (HH)-like model for the GnRH neuron is developed to reproduce each mode of burst firing with an appropriate set of conductances. Model outcomes for bursting are in agreement with the experimental recordings in terms of interburst interval, interspike interval, active phase duration, and other quantitative properties specific to each mode of bursting. The model also shows similar outcomes in membrane potential to those seen experimentally when tetrodotoxin (TTX) is used to block action potentials during bursting, and when estradiol transitions cells exhibiting slow oscillations to irregular bursting mode in vitro. Based on the parameter values used to reproduce each mode of bursting, the model suggests that GnRH neurons can switch between the two through changes in the maximum conductance of certain ionic currents, notably the slow inward Ca2+ current I s, and the Ca2+ -activated K+ current I KCa. Bifurcation analysis of the model shows that both modes of bursting are similar from a dynamical systems perspective despite differences in burst characteristics.










Similar content being viewed by others
References
Achard, P., & De Schutter, E. (2006). Complex parameter landscape for a complex neuron model. PLoS Computational Biology, 2, 0794–0804. doi:10.1371/journal.pcbi.0020094.
Amini, B., Clark, J. W., & Canavier, C. C. (1999). Calcium dynamics underlying pacemaker-like and burst firing oscillations in midbrain dopaminergic neurons: a computational study. Journal of Neurophysiology, 82, 2249–2261.
Bekkers, J. M. (2000). Properties of voltage-gated potassium currents in nucleated patches from large layer 5 cortical pyramidal neurons of the rat. Journal of Physiology, 525(Pt 3), 593–609. doi:10.1111/j.1469-7793.2000.t01-1-00593.x.
Belchetz, P. E., Plant, T. M., Nakai, Y., et al. (1978). Hypophysial responses to continuous and intermittent delivery of hypopthalamic gonadotropin-releasing hormone. Science, 202, 631–633. doi:10.1126/science.100883.
Bertram, R., Butte, M. J., Kiemel, T., & Sherman, A. (1995). Topological and phenomenological classification of bursting oscillations. Bulletin of Mathematical Biology, 57, 413–439. doi:10.1007/BF02460633.
Canavier, C. C., Clark, J. W., & Byrne, J. H. (1991). Simulation of the bursting activity of neuron R15 in Aplysia: role of ionic currents, calcium balance, and modulatory transmitters. Journal of Neurophysiology, 66, 2107–2124.
Carmeliet, E. (1987). Voltage-dependent block by tetrodotoxin of the sodium channel in rabbit cardiac Purkinje fibers. Biophysical Journal, 51, 109–114. doi:10.1016/S0006-3495(87)83315-5.
Chen X., Sneyd J. (2014). A Computational Model of the Dendron of the GnRH Neuron. Bulletin of Mathematical Biology 904–926. doi: 10.1007/s11538-014-0052-6.
Chen, X., Iremonger, K., Herbison, A., et al. (2013). Regulation of electrical bursting in a spatiotemporal model of a GnRH neuron. Bulletin of Mathematical Biology, 75, 1941–1960. doi:10.1007/s11538-013-9877-7.
Christian, C. A., & Moenter, S. M. (2010). The neurobiology of preovulatory and estradiol-induced gonadotropin- releasing hormone surges. Endocrine Reviews, 31, 544–577. doi:10.1210/er.2009-0023.
Chu, Z., & Moenter, S. M. (2006). Physiologic regulation of a tetrodotoxin-sensitive sodium influx that mediates a slow afterdepolarization potential in gonadotropin-releasing hormone neurons: possible implications for the central regulation of fertility. Journal of Neuroscience, 26, 11961–11973. doi:10.1523/JNEUROSCI.3171-06.2006.
Chu, Z., Andrade, J., Shupnik, M. A., & Moenter, S. M. (2009). Differential regulation of gonadotropin-releasing hormone neuron activity and membrane properties by acutely applied estradiol: dependence on dose and estrogen receptor subtype. Journal of Neuroscience, 29, 5616–5627. doi:10.1523/JNEUROSCI.0352-09.2009.
Chu, Z., Takagi, H., & Moenter, S. M. (2010). Hyperpolarization-activated currents in gonadotropin-releasing hormone (GnRH) neurons contribute to intrinsic excitability and are regulated by gonadal steroid feedback. Journal of Neuroscience, 30, 13373–13383. doi:10.1523/JNEUROSCI.1687-10.2010.
Chu, Z., Tomaiuolo, M., Bertram, R., & Moenter, S. M. (2012). Two types of burst firing in gonadotrophin-releasing hormone neurones. Journal of Neuroendocrinology, 24, 1065–1077. doi:10.1111/j.1365-2826.2012.02313.x.
Csercsik, D., Farkas, I., Hrabovszky, E., & Liposits, Z. (2012). A simple integrative electrophysiological model of bursting GnRH neurons. Journal of Computational Neuroscience, 32, 119–136. doi:10.1007/s10827-011-0343-y.
DeFazio, R. A., & Moenter, S. M. (2002). Estradiol feedback alters potassium currents and firing properties of gonadotropin-releasing hormone neurons. Molecular Endocrinology, 16, 2255–2265. doi:10.1210/me.2002-0155.
Destexhe, A., Mainen, Z. F., & Sejnowski, T. J. (1994). Synthesis of models for excitable membranes, synaptic transmission and neuromodulation using a common kinetic formalism. Journal of Computational Neuroscience, 1, 195–230. doi:10.1007/BF00961734.
Dickson, C. T., Magistretti, J., Shalinsky, M. H., et al. (2000). Properties and role of I(h) in the pacing of subthreshold oscillations in entorhinal cortex layer II neurons. Journal of Neurophysiology, 83, 2562–2579.
Doedel, E. J., & Oldeman, B. E. (2007). AUTO-07P : continuation and bifurcation software for ordinary differential equations. Montreal, Canada: Concordia University. http://indy.cs.concordia.ca/auto/. Accessed 1 Sep 2015.
Duan, W., Lee, K., Herbison, A. E., & Sneyd, J. (2011). A mathematical model of adult GnRH neurons in mouse brain and its bifurcation analysis. Journal of Theoretical Biology, 276, 22–34. doi:10.1016/j.jtbi.2011.01.035.
Dutton, A., & Dyball, R. E. (1979). Phasic firing enhances vasopressin release from the rat neurohypophysis. Journal of Physiology, 290, 433–440.
Ermentrout, B. (2002). Simulating, analyzing, and animating dynamical systems: a guide to XPPAUT for researchers and students. Philadelphia: SIAM.
Fletcher, P. A., & Li, Y. X. (2009). An integrated model of electrical spiking, bursting, and calcium oscillations in GnRH neurons. Biophysical Journal, 96, 4514–4524. doi:10.1016/j.bpj.2009.03.037.
Fox, R. F., & Lu, Y. N. (1994). Emergent collective behavior in large numbers of globally coupled independently stochastic ion channels. Physical Review E, 49, 3421–3431. doi:10.1103/PhysRevE.49.3421.
Glanowska, K. M., & Moenter, S. M. (2015). Differential regulation of GnRH secretion in the preoptic area (POA) and the median eminence (ME) in male mice. Endocrinology, 156, 231–241. doi:10.1210/en.2014-1458.
Hemond, P. J., O’Boyle, M. P., Roberts, C. B., et al. (2012). Simulated GABA synaptic input and L-type calcium channels form functional microdomains in hypothalamic gonadotropin-releasing hormone neurons. Journal of Neuroscience, 32, 8756–8766. doi:10.1523/JNEUROSCI.4188-11.2012.
Herbison, A. E. (1998). Multimodal influence of estrogen upon gonadotropin-releasing hormone neurons. Endocrine Reviews, 19, 302–330.
Hibino, H., Inanobe, A., Furutani, K., et al. (2010). Inwardly rectifying potassium channels : their structure, function, and physiological roles. Physiology Reviews, 90, 291–366. doi:10.1152/physrev.00021.2009.
Hiraizumi, Y., Nishimura, I., Ishii, H., et al. (2008). Rat GnRH neurons exhibit large conductance voltage- and Ca2 + −Activated K+ (BK) currents and express BK channel mRNAs. Journal of Physiological Sciences, 58, 21–29. doi:10.2170/physiolsci.RP013207.
Huguenard, J. R., & McCormick, D. A. (1992). Simulation of the currents involved in rhythmic oscillations in thalamic relay neurons. Journal of Neurophysiology, 68, 1373–1383.
Hunter, J. D. (2007). Matplotlib: a 2D graphics environment. Computing in Science and Engineering, 9, 99–104. doi:10.1109/MCSE.2007.55.
Izhikevich, E. M. (2000). Neural excitability, spiking and bursting. International Journal of Bifurcation and Chaos, 10, 1171–1266. doi:10.1142/S0218127400000840.
Kato, M., Ui-Tei, K., Watanabe, M., & Sakuma, Y. (2003). Characterization of voltage-gated calcium currents in gonadotropin-releasing hormone neurons tagged with green fluorescent protein in rats. Endocrinology, 144, 5118–5125. doi:10.1210/en.2003-0213.
Kuo, C. C., & Bean, B. P. (1994). Na + channels must deactivate to recover from inactivation. Neuron, 12, 819–829. doi:10.1016/0896-6273(94)90335-2.
LeBeau, A. P., Van Goor, F., Stojilkovic, S. S., & Sherman, A. (2000). Modeling of membrane excitability in gonadotropin-releasing hormone-secreting hypothalamic neurons regulated by Ca2 + −mobilizing and adenylyl cyclase-coupled receptors. Journal of Neuroscience, 20, 9290–9297.
Lee, K., Duan, W., Sneyd, J., & Herbison, A. E. (2010). Two slow calcium-activated afterhyperpolarization currents control burst firing dynamics in gonadotropin-releasing hormone neurons. Journal of Neuroscience, 30, 6214–6224. doi:10.1523/JNEUROSCI.6156-09.2010.
Lee, K., Liu, X., & Herbison, A. E. (2012). Burst firing in gonadotrophin-releasing hormone neurones does not require ionotrophic GABA or glutamate receptor activation. Journal of Neuroendocrinology, 24, 1476–1483. doi:10.1111/j.1365-2826.2012.02360.x.
LeMasson G., Maex R. (2001). Introduction to equation solving and parameter Fitting. In E. De Schutter (Ed.), Computational neuroscience: realistic modeling for experimentalists (pp. 1–25). CRC Press.
Liu, X., & Herbison, A. E. (2008). Small-conductance calcium-activated potassium channels control excitability and firing dynamics in Gonadotropin-Releasing Hormone (GnRH) neurons. Endocrinology, 149, 3598–3604. doi:10.1210/en.2007-1631.
Longtin, A. (1997). Autonomous stochastic resonance in bursting neurons. Physical Review E, 55, 868–876. doi:10.1103/PhysRevE.55.868.
Longtin, A., Laing, C., & Chacron, M. J. (2003). Correlations and memory in neurodynamical systems. In G. Rangarajan & M. Ding (Eds.), Processes with long-range correlations (pp. 286–308). Berlin: Springer.
Lu, B., Su, Y., Das, S., et al. (2007). The neuronal channel NALCN contributes resting sodium permeability and is required for normal respiratory rhythm. Cell, 129, 371–383. doi:10.1016/j.cell.2007.02.041.
Magistretti, J., & Alonso, A. (1999). Biophysical properties and slow voltage-dependent inactivation of a sustained sodium current in entorhinal cortex layer-II principal neurons: a whole-cell and single-channel study. The Journal of General Physiology, 114, 491–509. doi:10.1085/jgp.114.4.491.
Miyasho, T., Takagi, H., Suzuki, H., et al. (2001). Low-threshold potassium channels and a low-threshold calcium channel regulate Ca2+ spike firing in the dendrites of cerebellar Purkinje neurons: a modeling study. Brain Research, 891, 106–115. doi:10.1016/S0006-8993(00)03206-6.
Moenter, S. M. (2010). Identified GnRH neuron electrophysiology: a decade of study. Brain Research, 1364, 10–24. doi:10.1016/j.brainres.2010.09.066.
Moenter, S. M., Chu, Z., & Christian, C. A. (2009). Neurobiological mechanisms underlying oestradiol negative and positive feedback regulation of gonadotrophin-releasing hormone neurones. Journal of Neuroendocrinology, 21, 327–333. doi:10.1111/j.1365-2826.2009.01826.x.
Nunemaker, C. S., Defazio, R. A., & Moenter, S. M. (2002). Episodic firing patterns of GnRH neurons. Endocrinology, 143, 2284–2292.
Nunemaker, C. S., Straume, M., Defazio, R. A., & Moenter, S. M. (2003). Gonadotropin-releasing hormone neurons generate interacting rhythms in multiple time domains. Endocrinology, 144, 823–831. doi:10.1210/en.2002-220585.
Pielecka, J., & Moenter, S. M. (2006). Effect of steroid milieu on gonadotropin-releasing hormone-1 neuron firing pattern and luteinizing hormone levels in male mice. Biology of Reproduction, 74, 931–937. doi:10.1095/biolreprod.105.049619.
Pielecka-Fortuna, J., DeFazio, R. A., & Moenter, S. M. (2011). Voltage-gated potassium currents are targets of diurnal changes in estradiol feedback regulation and kisspeptin action on gonadotropin-releasing hormone neurons in mice. Biology of Reproduction, 85, 987–995. doi:10.1095/biolreprod.111.093492.
Plant, R. E. (1981). Bifurcation and resonance in a model for bursting nerve cells. Journal of Mathematical Biology, 11, 15–32. doi:10.1007/BF00275821.
Rinzel, J., & Lee, Y. S. (1987). Dissection of a model for neuronal parabolic bursting. Journal of Mathematical Biology, 25, 653–675. doi:10.1007/BF00275501.
Roberts, C. B., O’Boyle, M. P., & Suter, K. J. (2009). Dendrites determine the contribution of after depolarization potentials (ADPs) to generation of repetitive action potentials in hypothalamic gonadotropin releasing-hormone (GnRH) neurons. Journal of Computational Neuroscience, 26, 39–53. doi:10.1007/s10827-008-0095-5.
Sun, J., Chu, Z., & Moenter, S. M. (2010). Diurnal in vivo and rapid in vitro effects of estradiol on voltage-gated calcium channels in gonadotropin-releasing hormone neurons. Journal of Neuroscience, 30, 3912–3923. doi:10.1523/JNEUROSCI.6256-09.2010.
Van Geit, W., Achard, P., & De Schutter, E. (2007). Neurofitter: a parameter tuning package for a wide range of electrophysiological neuron models. Frontiers in Neuroinformatics, 1, 1. doi:10.3389/neuro.11.001.2007.
Van Goor, F., LeBeau, A. P., Krsmanovic, L. Z., et al. (2000). Amplitude-dependent spike-broadening and enhanced Ca(2+) signaling in GnRH-secreting neurons. Biophysical Journal, 79, 1310–1323. doi:10.1016/S0006-3495(00)76384-3.
Wang, Y., Garro, M., & Kuehl-Kovarik, M. C. (2010). Estradiol attenuates multiple tetrodotoxin-sensitive sodium currents in isolated gonadotropin-releasing hormone neurons. Brain Research, 1345, 137–145. doi:10.1016/j.brainres.2010.05.031.
Wildt, L., Häusler, A., Marshall, G., et al. (1981). Frequency and amplitude of gonadotropin-releasing hormone stimulation and gonadotropin secretion in the rhesus monkey. Endocrinology, 109, 376–385. doi:10.1210/endo-109-2-376.
Willms, A. R. (2002). NEUROFIT: software for fitting Hodgkin-Huxley models to voltage-clamp data. Journal of Neuroscience Methods, 121, 139–150. doi:10.1016/S0165-0270(02)00227-3.
Willms, A. R., Baro, D. J., Harris-Warrick, R. M., & Guckenheimer, J. (1999). An improved parameter estimation method for Hodgkin-Huxley models. Journal of Computational Neuroscience, 6, 145–168. doi:10.1023/A:1008880518515.
Wintermantel, T. M., Campbell, R. E., Porteous, R., et al. (2006). Definition of estrogen receptor pathway critical for estrogen positive feedback to gonadotropin-releasing hormone neurons and fertility. Neuron, 52, 271–280. doi:10.1016/j.neuron.2006.07.023.
Zhang, C., Bosch, M. A., Rick, E. A., et al. (2009). 17Beta-estradiol regulation of T-type calcium channels in gonadotropin-releasing hormone neurons. Journal of Neuroscience, 29, 10552–10562. doi:10.1523/JNEUROSCI.2962-09.2009.
Acknowledgments
This work was supported by the Natural Sciences and Engineering Council of Canada (NSERC) discovery grant to AK and NIH grants R01HD34860 and R01HD41469 to SMM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Action Editor: David Terman
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 632 kb)
Rights and permissions
About this article
Cite this article
Moran, S., Moenter, S.M. & Khadra, A. A unified model for two modes of bursting in GnRH neurons. J Comput Neurosci 40, 297–315 (2016). https://doi.org/10.1007/s10827-016-0598-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-016-0598-4