Abstract
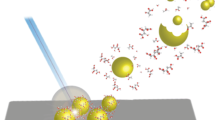
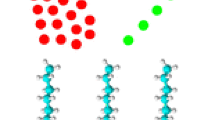
Self-assembled monolayers (SAMs) on coinage metal provide versatile modeling systems for studies of interfacial electron transfer, biological interactions, molecular recognition and other interfacial phenomena. Recently the bonding of enzyme to SAMs of alkanethiols onto Au electrode surfaces was exploited to produce a bio-sensing system. In this work, the attachment of trypsin to a SAMs surface of 11-mercaptoundecanoic acid was achieved using water soluble N-ethyl-N ′-(3-dimethylaminopropyl)carbodiimide hydrochloride and N-hydroxysuccinimide as coupling agent. The thickness of SAMs was determined by optical ellipsometer; contact angles of the modified Au surfaces were measured in air using a goniometer. The Second Harmony Generation data displays the last few percents of the alkylthiol molecules adsorbed and produced the complete monolayer by inducing the transition from a high number of gauche defects to an all-trans conformation. Using X-ray Photoelectron Spectroscopy (XPS) and Fourier-Transformed Infrared Reflection-Absorption and Attenuated Total Reflection Spectroscopes (FTIR-RAS and ATR), we examined the chemical structures of samples with different treatments. By matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS), we demonstrated the digestion of bovine serum albumin (BSA) on the trypsin-immobilized SAMs surface.
Experimental results have revealed that the XPS C1s core levels at 286.3 and 286.5 eV (Amine bond), 288.1 eV (Amide bond) and 289.3 eV (Carboxylic acid) illustrate the immobilization of trypsin. These data were also in good agreement with FTIR-ATR spectra for the peaks valued at 1659.4 cm− 1 (Amide I) and 1546.6 cm− 1 (Amide II). Using MALDI-TOF MS observations, analytical results have demonstrated the BSA digestion of the immobilized trypsin on the functionalized SAMs surface. For such surfaces, BSA was digested on the trypsin-immobilized SAMs surface, which shows the enzyme digestion ability of the immobilized trypsin. The terminal groups of the SAMs structure can be further functionalized with biomolecules or antibodies to develop surface-base diagnostics, biosensors, or biomaterials.
Similar content being viewed by others
References
J. C. HORNE and G. J. BLANCHARD, J. Am. Chem. Soc. 120 (1998) 6336.
F. MORHARD, J. SCHUMACHER, A. LENENBACH, T. WILHELM, R. DAHINT, M. GRUNZE and D. S. EVERHART, Proc. - Electrochem. Soc. 97(19) (1997) 1058.
K. BIERBAUM, M. GRUNZE, A. BASKI, L. F. CHI, W. SCHREPP and H. FUCHS, Langmuir 11 (1995) 2143.
A. SCHERTEL, C. WOLL and M. GRUNZE, J. Phys. IV. 7 (1997) 537.
C. YAN, M. ZHARNIKOV, A. GOELZHAEUSER and M. GRUNZE, Langmuir 16 (2000) 6208.
H. J. HIMMEL, K. WEISS, B. JAEGER, O. DANNENBERGER, M. GRUNZE and C. WOELL, Langmuir 13 (1997) 4943.
C. JUNG, O. DANNENBERGER, Y. XU, M. BUCK and M. GRUNZE, Langmuir 14 (1998) 1103.
R. G. NUZZO and D. L. ALLARA, J. Am. Chem. Soc. 105 (1983) 4481.
C. W. SHEEN, J. X. SHI, J. MARTENSSON, A. N. PARIKH and D. L. ALLARA, J. Am. Chem. Soc. 114 (1992) 1514.
Y. GU, Z. LIN, R. A. BUTERA, V. S. SMENTKOWSKI and D. H. WALDECK, Langmuir 11 (1995) 1849.
T. J. GARDNER, C. D. FRISBIE and M. S. WRIGHTON, J. Am. Chem. Soc. 117 (1995) 6927.
C. D. BAIN, E. B. TROUGHTON, Y. TAO, J. EVALL, G. M. WHITESIDES and R. G. NUZZO, J. Am. Chem. Soc. 111 (1989) 321.
A. ULMAN, in “An Introduction to Ultrathin Origanic Films” (Academic Press, San Diego, 1991).
L. H. DUBOIS and R. G. NUZZO, Annu. Rev. Phy. Chem. 43 (1992) 437.
A. ULMAN, Chem. Rev. 96 (1996) 1533.
P. E. LAIBINIS, R. G. NUZZO and G. M. WHITESIDES, J. Phys. Chem. 96 (1992) 5097.
S. A. JOYCE, R. C. THOMAS, J. E. HOUSTON, T. A. MICHALSKE and R. M. CROOKS, Phys. Rev. Lett. 68 (1992) 2790.
J. J. GOODING and D. B. HIBBERT, Trends in Analytical Chemistry 18 (1999) 525.
C. ERDELEN, L. HäUSSLING, R. NAUMANN, H. RINGSDORF, H. WOLF, J. YANG, M. LILEY, J. SPINKE and W. KNOLL, Langmuir 10 (1994) 1246.
P. E. LAIBINIS and G. M. WHITESIDES, J. Am. Chem. Soc. 114 (1992) 9022.
D. G. BUERK, in “Biosensor: Theory and Applications” (Tech. Pub. Comp. Inc, 1993).
I. K. KANG, B. K. KWON, J. H. LEE and H. B. LEE, Biomaterials 14 (1993) 787.
Y. C. TYAN, J. D. LIAO, R. KLAUSER, I. D. WU and C. C. WENG, Biomaterials 23 (2002) 65.
O. DANNENBERGER, M. BUCK and M. GRUNZE, J. Phys. Chem. B. 103 (1999) 2202.
B. L. FREY and R. M. CORN, Anal. Chem. l 68 (1996) 3187.
E. L. SMITH, C. A. ALVES, J. W. ANDEREGG, M. D. PORTER and L. M. SIPERKO, Langmuir 8 (1992) 2707.
L. SUN, R. M. CROOKS and A. J. RICCO, Langmuir 9 (1993) 1775.
Y. MIURA, S. KIMURA, Y. IMANISHI and J. UMEMURA, Langmuir 15 (1999) 1155.
C. J. DELDEN, J. P. LENS, R. P. H. KOOYMAN, G. H. M. ENGBERS and J. FEIJEN, Biomaterials 18 (1997) 845.
A. J. KUIJPERS, P. B. WACHEM, M. J. A. LUYN, L. A. BROUWER, G. H. M. ENGBERS, J. KRIJGSVELD, S. A. J. ZAAT, J. DANKERT and J. FEIJEN, Biomaterials 21 (2000) 1763.
M. LESTELIUS, B. LIEDBERG and P. TENGVALL, Langmuir 13 (1997) 5900.
A. R. NOBLE-LUGINBUHI and R. G. NUZZO, Langmuir 17 (2001) 3937.
C. E. D. CHIDSEY and D. N. LOIACONO, Langmuir 6 (1990) 682.
P. E. LAIBINIS, G. M. WHITESIDES, D. L. ALLARA, Y. T. TAO, A. N. PARIKH and R. G. NUZZO, J. Am. Chem. Soc. 113 (1991) 7152.
Y. ITO, M. KAJIHARA and Y. IMANISHI, J. Biomed. Mater. Res. 25 (1991) 1325.
Y. C. TYAN, J. D. LIAO, S. P. LIN and C. C. CHEN, J. Biomed. Mater. Res. 67A (2003) 1033.
J. F. MOULDER, W. F. STICKLE, P. E. SOBOL and K. D. BOMBEN, in “Handbook of X-ray Photoelectron Spectroscopy” (Physical Electronics, Inc. Minnesota, 1995).
G. BEAMSON and D. BRIGGS, in “High Resolution XPS of Organic Polymers. The Scienta ESCA300 Database” (Wiley & Sons Inc., New York, 1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tyan, YC., Liao, JD., Jong, SB. et al. Characterization of trypsin immobilized on the functionable alkylthiolate self-assembled monolayers: A preliminary application for trypsin digestion chip on protein identification using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J Mater Sci: Mater Med 16, 135–142 (2005). https://doi.org/10.1007/s10856-005-5987-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10856-005-5987-6