Abstract
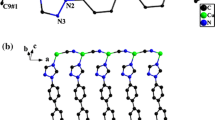
The structure of the 3D-supramolecular coordination polymer (SCP) 3∞ {[CuΙ(CN)·2(phen)·CuΙΙ(CN)2·(phen)]·5H2O}, 1 contains the [CuΙ(CN)·2(phen)], [CuΙΙ(CN)2·(phen)] molecular complexes and five water molecules which are connected with each other by hydrogen bonds. The most intriguing feature in the structure of 1 is the unusual supramolecular interactions, including hydrogen bonds which involve the terminal cyanide and the phenanthroline (phen) ligands and π–π stacking which generate a unique 3D-supramolecular architecture in the solid state. The emission spectrum of 1 displays a band centered at 420 nm which was assigned to So-T transition in the CuCN fragments. This band suffers a shift to longer wavelength by about 30 nm than that of CuCN itself. The emission bands at 435, 450, and 475 correspond to the close laying π–π* transitions of the coordinated phen. The main band at 435 nm exhibits a red shift than that of phen by about 70 nm which makes it attractive as luminescent sensor. Also, the SCP 1 was used as heterogeneous catalyst for the degradation of Metanil Yellow (MY) dye by dilute solution of hydrogen peroxide as oxidant. The reaction is first order with respect to the MY dye. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was used to determine the in vitro antitumor activity of the SCP 1 on human breast cancer cell line, MCF7. The cytotoxicity of the SCP 1 is more efficient than that of the Doxorubicin (DOX) drug. The thermal stability has been also investigated.











Similar content being viewed by others
References
B.A. Maynard, R.E. Sykora, J.T. Maguec, A.E.V. Gorden, Chem. Commun. 46, 4944 (2010)
J. Larionova, Y. Guari, C. Blanc, P. Dieudonne, A. Tokarev, C. Guerin, Langmuir 25, 1138 (2009)
Z. Su, K. Cai, J. Fan, S.S. Chen, M.S. Chen, W.Y. Sun, Cryst. Eng. Commun. 12, 100 (2010)
L.F. Ma, L.Y. Wang, M. Du, S.R. Batton, Inorg. Chem. 49, 365 (2010)
S.E.H. Etaiw, A.S. Sultan, M.M. El-Bendary, J. Organomet. Chem. 696, 1668 (2011)
M. Ohba, H. Okawa, Coord. Chem. Rev. 198, 313 (2000)
L.M. Toma, R. Lescouezec, D. Cangussu, R. Llusar, J. Mata, S. Spey, J.A. Thomas, F. Lloret, M. Julve, Inorg. Chem. Commun. 8, 382 (2005)
S.E.H. Etaiw, S.A. Amer, M.M. El-Bendary, J. Mater. Sci. 45, 1307 (2010)
S.E.H. Etaiw, S.A. Amer, M.M. El-Bendary, Polyhedron 28, 2385 (2009)
S.E.H. Etaiw, M.M. El-Bendary, J. Inorg. Organomet. Polym. 20, 739 (2010)
A.N. Ley, L.E. Dunaway, T.P. Brewster, M.D. Dembo, T.D. Harris, F. Baril-Robert, X. Li, H.H. Patterson, R.D. Pike, Chem. Commun. 46, 4565 (2010)
S.V. Wegner, H. Arslan, M. Sunbul, J. Yin, C. He, J. Am. Chem. Soc. 132, 2567 (2010)
E. Colacio, R. Kivekas, F. Lloret, M. Sunberg, J.S. Varela, M. Bardaji, A. Laguna, Inorg. Chem. 41, 5141 (2002)
L.-L. Li, L.-L. Liu, A.-X. Zheng, Y.-J. Chang, M. Dai, Z.-G. Ren, H.-X. Lia, J.-P. Lang, Dalton Trans. 39, 7659 (2010)
L. Hou, W.-J. Shi, Y.-Y. Wang, B. Liu, W.-H. Huang, Q.-Z. Shi, CrystEngComm 12, 4365 (2010)
X. He, C.Z. Lu, C.D. Wu, L.J. Chen, Eur. J. Inorg. Chem. 12, 2491 (2006)
S.E.H. Etaiw, A.S. Badr El-din, J. Inorg. Organomet. Polym. 21, 110 (2011)
C. Kappestein, U. Schubert, Chem. Commun. 1116 (1980)
K. Wasielewski, R. Mattes, Z. Naturforsch. B Chem. Sci. 47, 1795 (1992)
P.G. Sammes, G. Yahioglu, Chem. Soc. Rev. 23, 327 (1994)
J.-P. Sauvage, C.O. Dietrich-Buchecker (ed.), Molecular catenanes, rotaxanes and knots. A journey through the world of molecular topology (Wiley-VCH, Weinheim, Germany, 1999)
S. Bonnet, J.P. Collin, M. Koizumi, P. Mobian, J.P. Sauvage, Adv. Mater. 18, 1239 (2006)
K. Hayashi, H. Akutsu, H. Ozaki, H. Sawai, Chem. Commun. 1386 (2004)
J.W. Eastes, W.M. Burgess, J. Am. Chem. Soc. 64, 1187 (1942)
M. Wicholas, T. Wolford, Inorg. Chem. 13, 316 (1974)
M. Dunaj-Jurčo, I. Potočňák, J. Cíbik, M. Kabešová, Acta Cryst. C49, 1479 (1993)
H. Masuda, K. Machida, M. Munakata, S. Kitagawa, H. Shimono, J. Chem. Soc. Dalton Trans. 1907 (1988)
L. Stamp, T. Dieck, Inorg. Chim. Acta 129, 107 (1987)
T. Pintauer, J. Organomet. Chem. 691, 3948 (2006)
R.H. Holm, P. Kennepohl, E.I. Solomon, Chem. Rev. 96, 2239 (1996)
M. Kondo, Y. Shibuya, K. Nabari, M. Miyazawa, S. Yasue, K. Maeda, F. Uchida, Inorg. Chem. Commun. 10, 1311 (2007)
J. Costamagna, F. Caruso, J. Vargas, V. Manriquez, Inorg. Chim. Acta 267, 151 (1998)
J.R.B. Gomes, M.A.V. Ribeiro da Silva, Inorg. Chem. Commun. 6, 149 (2003)
L. Gutierrez, G. Alzuet, J. Borrás, M. Liu-González, F. Sanz, A. Castiňeiras, Polyhedron 20, 703 (2001)
V. Broughton, G. Bernardinelli, A.F. Williams, Inorg. Chim. Acta 275–276, 279 (1998)
K.R. Dunbar, R.A. Heintz, Prog. Inorg. Chem. 45, 283 (1997)
K.-Y. Ho, W.-Y. Yu, K.-K. Cheung, C.-M. Che, Chem. Commun. 2101 (1998)
X.-C. Huang, S.-L. Zheng, J. -P. Zhang, X.-M. Chen, Eur. J. Inorg. Chem. 1024 (2004)
A. Spalleti, G. Cruciani, U. Mazzucato, J. Mol. Struct. 612, 339 (2002)
H.H. Jalfé, M. Orechin, Theory and Applications of Ultraviolet Spectroscopy, 5th edn. (Wiley, New York, 1970)
N. Armaroli, L.D. Cola, V. Balzani, J.-P. Sauvage, C. Dietrich-Buchecker, J.-M. Kern, J. Chem. Soc. Faraday Trans. 88, 553 (1992)
B.N. Bandyopadhyay, A. Harriman, J. Chem. Soc. Faraday Trans. 1, 73 (1977)
G. M. Badger, I. S. Walker, J. Chem. Soc. 122 (1956)
K. Yamamote, T. Takemura, H. Baba, Bull. Chem. Soc. Jpn. 51, 729 (1978). and references therein
M.J. Lim, C.A. Murray, T.A. Tronic, K.E. deKrafft, A.N. Ley, J.C. deButts, R.D. Pike, H. Lu, H.H. Patterson, Inorg. Chem. 47, 6931 (2008)
C.A. Bayse, T.P. Brewster, R.D. Pike, Inorg. Chem. 48, 174 (2009)
J.M. Monteagudo, A. Durάn, I. San Martin, M. Aguίrre, Appl. Catal. B Environ. 95, 120 (2010)
L. Dapeng, G. Jiuhui, J. Environ. Sci. 21, 713 (2009)
J.M. Campos-Martin, G. Blanco-Brieva, J.L.G. Fierro, Angew. Chem. Int. Ed. 45, 6962 (2006)
Y. Lu, M. Tonigod, B. Bredenkötter, D. Volkmer, J. Hitzbleck, G. Langstein, Z. Anorg, Allg. Chem. 634, 2411 (2008)
C. Flox, S. Ammar, C. Arias, E. Brillas, A. Vargas-Zaval, R. Abdehdi, Appl. Catal. B Environ. 67, 93 (2006)
J. De Laat, T.G. Le, Appl. Catal. B Environ. 66, 137 (2006)
G. Chelucci, R.P. Thummel, Chem. Rev. 102, 3129 (2002)
A.S. Sultan, J. Xie, M.J. LeBaron, L.E. Ealley, M.T. Nevalainen, H. Rui, Oncogene 24, 746 (2005)
A.S. Sultan, H. Brim, Z.A. Sherif, Cancer Sci. 2, 272 (2008)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Etaiw, S.Ed.H., Amer, S.A. & El-Bendary, M.M. A Mixed Valence Copper Cyanide 3D-supramolecular Coordination Polymer Containing 1,10-Phenathorline Ligand as a Potential Antitumor Agent, Effective Catalyst and Luminescent Material. J Inorg Organomet Polym 21, 662–672 (2011). https://doi.org/10.1007/s10904-011-9532-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-011-9532-4