Abstract
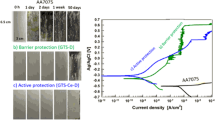
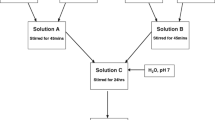
In sol-gel chemistry, hydrolysis is the key step in the formation of the reactive hydroxide groups that are responsible for the formation of inorganic networks via the occurrence of condensation reactions. Though previous studies have investigated the effect of the hydrolysis conditions on the structure of organically modified silicates (ormosils), no study, to our knowledge, has investigated this variable on the structure of hybrid materials prepared by combinations of an ormosil and a transition metal (TM). Here, we propose to investigate this effect in a hybrid material composed of 3-trimethoxysilylpropylmethacrylate and a zirconium complex. To also highlight the effects of the precursor’s concentrations on the hydrolysis and condensation reactions of the hybrid materials, their relative content was altered along with the hydrolysis degree. The anticorrosion barrier properties were identified by characterisation of coatings deposited on AA2024-T3 substrates and correlation between the structure and the anticorrosion properties of the coatings were performed based on results obtained from structural characterisations (DLS, FTIR, 29Si-NMR, DSC, AFM and SEM) and corrosion testing (EIS and NSS). It is demonstrated that competition in the formation of siloxane and Si-O-Zr bonds takes place and can be controlled by the degree of hydrolysis and the concentration of the zirconium complex. This effect was found to dramatically alter the morphology of the coatings and their subsequent anticorrosion performances. At short-term exposure times, it is found that the most condensed materials exhibited a higher corrosion resistance while over longer periods the performances were found to level. This article highlighted the critical impact of the hydrolysis degree and zirconium concentration on the connectivity of hybrid sol-gel coatings and the impact this has on corrosion performances.
















Similar content being viewed by others
References
Official Journal of the European Union “Commission Directive 2011/37/EU”, OJ L 269
EPA Federal Register (1995) National Emission Standards for Hazardous Air Pollutants for Source Categories: Aerospace Manufacturing and Rework Facilities, 60:45947
Pommiers S, Frayret J, Castetbon A, Potin-Gautier M (2014) Corros Sci 84:135–146
Hornberger H, Virtanen S, Boccaccini AR (2012) Acta Biomater 8:2442–2455
Amini R, Sarabi AA (2011) Appl Surf Sci 257:7134–7139
Figueira RB, Fontinha IR, Silva CJR, Pereira EV (2016) Coatings 6(1):12
Amiri S, Rahimi A (2016) Iran Polym J 25:559–577
Marques ME, Mansur AAP, Mansur HS (2013) Appl Surf Sci 275:347–360
Su H-Y, Chen P-L, Lina C-S (2016) Corros Sci 102:63–71
Chichkov BN, Farsari M (2009) Nat Photonics 3:450–452
Oubaha M, Gorin A, McDonagh C, Duffy B, Copperwhite R (2015) Sens Actuators B 221:96–103
Fedel M, Callone E, Fabbian M, Deflorian F, Dirè S (2017) Appl Surf Sci 414:82–91
Moutarlier V, Neveu B, Gigandet MP (2008) Surf Coat Technol 202:2052
Upadhyay V, Bergseth Z, Kelly B, Battocchi D (2017) Coatings 7:86
J Colreavy, B Duffy, PCR Varma, H Hayden, M Oubaha Patent “Sol-gel coating compositions and their process of preparation” EP 2220176 A2
Lebeau B, Innocenzi P (2011) Chem Soc Rev 40:886–906
Ferreira RAS, André PS, Carlos LD (2010) Opt Mater 32:1397–1409
Elmaghrum S, Gorin A, Kribich RK, Corcoran B, Copperwhite R, McDonagh C, Oubaha M (2013) Sens Actuators B 177:357–363
Oubaha M, Kavanagh A, Gorin A, Bickauskaite G, Byrne R, Farsari M, Winfield R, Diamond Dermot, McDonagh Colette, Copperwhite Robert (2012) J Mater Chem 22:10552
Delattre L, Dupuy C, Babonneau F (1994) Characterization of the hydrolysis and polymerization processes of methacryloxypropyltrimethoxysilane - code: BP18. J Sol-Gel Sci Technol 2(1–3):185–188
Gualandris V, Babonneau F, Janicke MT, Chmelka BF (1998) NMR studies on hydrolysis and condensation reactions of alkoxysilanes containing Si-H bonds. J Sol-Gel Sci Technol 13:75–80
Babonneau F, Maquet J (2000) Nuclear magnetic resonance techniques for the structural characterization of siloxane-oxide hybrid materials. Polyhedron 19(3):315–322
Cullen M, Morshed M, O’Sullivan M, MacHugh E, Duffy B, Oubaha M (2017) J Sol-Gel Sci Technol 82(3):801
Copperwhite R, O’Sullivan M, Boothman C, Gorin A, McDonagh C, Oubaha M (2011) Microfluid Nanofluid 11:283–296
Morrow BA, McFarlan AJ (1992) J Phys Chem 96:1395–1400
Elvira MR, Alejandra Mazo M, Tamayo A, Rubio F, Rubio J, Oteo JL (2013) J Chem Eng 7:120–131
Pickup DM, Mountjoy G, Wallidge GW, Newport RJ, Smith ME (1999) Phys Chem Chem Phys 1:2527
Hanuhov T, Asulin E, Gvishi R (2017) J Non-Cryst Solids 471:301
Lerot L, Low PF (1976) Clays Clay Miner 24:191–199
Handke M, Kwaśny M (2014) Vib Spectrosc 74:127–131
Handke M, Kwas̈ny M (2014) Infrared spectroscopic study of octahydridooctasilsesquioxane hydrolytic polycondensation. Vib Spectrosc 74:127–131
Delattre L, Babonneau F (1997) Chem Mater 9:2385–2394
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Highlights
-
Conjugated effects of the hydrolysis degree and transition metal concentration on the structure and anticorrosion properties of hybrid sol-gel coatings are investigated.
-
Correlation between the structure and anticorrosion performances of hybrid sol-gel coatings is proposed. The structure of the materials is characterised by 29Si-NMR, FTIR, DLS, DSC, AFM and SEM, while the anticorrosion performances are identified by EIS and NSS.
-
The condensation of the coatings is found to be strongly relying on the hydrolysis degree and transition metal concentration.
-
The short term anticorrosion performances are optimum for highly condensed coatings, while long term resistances are similar regardless of the condensation of the coatings.
-
The nature of the coating/metal interface is dependent on the condensation of the materials.
Rights and permissions
About this article
Cite this article
Cullen, M., O’Sullivan, M., Kumar, A.M. et al. The role of the hydrolysis and zirconium concentration on the structure and anticorrosion performances of a hybrid silicate sol-gel coating. J Sol-Gel Sci Technol 86, 553–567 (2018). https://doi.org/10.1007/s10971-018-4657-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-018-4657-3