Abstract
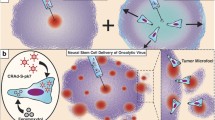
The malignant primary brain tumor, glioblastoma (GBM) is generally incurable. New approaches are desperately needed. Adeno-associated virus (AAV) vector-mediated delivery of anti-tumor transgenes is a promising strategy, however direct injection leads to focal transgene spread in tumor and rapid tumor division dilutes out the extra-chromosomal AAV genome, limiting duration of transgene expression. Intravenous (IV) injection gives widespread distribution of AAV in normal brain, however poor transgene expression in tumor, and high expression in non-target cells which may lead to ineffective therapy and high toxicity, respectively. Delivery of transgenes encoding secreted, anti-tumor proteins to tumor stromal cells may provide a more stable and localized reservoir of therapy as they are more differentiated than fast-dividing tumor cells. Reactive astrocytes and tumor-associated macrophage/microglia (TAMs) are stromal cells that comprise a large portion of the tumor mass and are associated with tumorigenesis. In mouse models of GBM, we used IV delivery of exosome-associated AAV vectors driving green fluorescent protein expression by specific promoters (NF-κB-responsive promoter and a truncated glial fibrillary acidic protein promoter), to obtain targeted transduction of TAMs and reactive astrocytes, respectively, while avoiding transgene expression in the periphery. We used our approach to express the potent, yet toxic anti-tumor cytokine, interferon beta, in tumor stroma of a mouse model of GBM, and achieved a modest, yet significant enhancement in survival compared to controls. Noninvasive genetic modification of tumor microenvironment represents a promising approach for therapy against cancers. Additionally, the vectors described here may facilitate basic research in the study of tumor stromal cells in situ.
Graphical abstract







Similar content being viewed by others
References
Dolecek TA, Propp JM, Stroup NE, Kruchko C (2012) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005–2009. Neuro Oncol 14(Suppl 5):v1–v49
Wakimoto H, Johnson PR, Knipe DM, Chiocca EA (2003) Effects of innate immunity on herpes simplex virus and its ability to kill tumor cells. Gene Ther 10(11):983–990
Harding TC, Lalani AS, Roberts BN, Yendluri S, Luan B, Koprivnikar KE, Gonzalez-Edick M, Huan-Tu G, Musterer R, VanRoey MJ, Ozawa T, LeCouter RA, Deen D, Dickinson PJ, Jooss K (2006) AAV serotype 8-mediated gene delivery of a soluble VEGF receptor to the CNS for the treatment of glioblastoma. Mol Ther 13(5):956–966
Wollmann G, Tattersall P, van den Pol AN (2005) Targeting human glioblastoma cells: comparison of nine viruses with oncolytic potential. J Virol 79(10):6005–6022
Harding TC, Dickinson PJ, Roberts BN, Yendluri S, Gonzalez-Edick M, Lecouteur RA, Jooss KU (2006) Enhanced gene transfer efficiency in the murine striatum and an orthotopic glioblastoma tumor model, using AAV-7- and AAV-8-pseudotyped vectors. Hum Gene Ther 17(8):807–820
Hadaczek P, Mirek H, Berger MS, Bankiewicz K (2005) Limited efficacy of gene transfer in herpes simplex virus-thymidine kinase/ganciclovir gene therapy for brain tumors. J Neurosurg 102(2):328–335
Maguire CA, Meijer DH, LeRoy SG, Tierney LA, Broekman ML, Costa FF, Breakefield XO, Stemmer-Rachamimov A, Sena-Esteves M (2008) Preventing growth of brain tumors by creating a zone of resistance. Mol Ther 16(10):1695–1702
Meijer DH, Maguire CA, LeRoy SG, Sena-Esteves M (2009) Controlling brain tumor growth by intraventricular administration of an AAV vector encoding IFN-beta. Cancer Gene Ther 16(8):664–671
Crommentuijn MH, Maguire CA, Niers JM, Vandertop WP, Badr CE, Wurdinger T, Tannous BA (2015) Intracranial AAV-sTRAIL combined with lanatoside C prolongs survival in an orthotopic xenograft mouse model of invasive glioblastoma. Mol Oncol 10(4):625–634
Hicks MJ, Funato K, Wang L, Aronowitz E, Dyke JP, Ballon DJ, Havlicek DF, Frenk EZ, De BP, Chiuchiolo MJ, Sondhi D, Hackett NR, Kaminsky SM, Tabar V, Crystal RG (2015) Genetic modification of neurons to express bevacizumab for local anti-angiogenesis treatment of glioblastoma. Cancer Gene Ther 22(1):1–8
Foust KD, Nurre E, Montgomery CL, Hernandez A, Chan CM, Kaspar BK (2009) Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes. Nat Biotechnol 27(1):59–65
Ahmed SS, Li H, Cao C, Sikoglu EM, Denninger AR, Su Q, Eaton S, Liso Navarro AA, Xie J, Szucs S, Zhang H, Moore C, Kirschner DA, Seyfried TN, Flotte TR, Matalon R, Gao G (2013) A single intravenous rAAV injection as late as P20 achieves efficacious and sustained CNS Gene therapy in Canavan mice. Mol Ther 21(12): 2136–2147
Calcedo R, Vandenberghe LH, Gao G, Lin J, Wilson JM (2009) Worldwide epidemiology of neutralizing antibodies to adeno-associated viruses. J Infect Dis 199(3):381–390
Glass R, Synowitz M (2014) CNS macrophages and peripheral myeloid cells in brain tumours. Acta Neuropathol 128(3):347–362
Okolie O, Bago JR, Schmid RS, Irvin DM, Bash RE, Miller CR, Hingtgen SD (2016) Reactive astrocytes potentiate tumor aggressiveness in a murine glioma resection and recurrence model. Neuro Oncol 18(12):1622–1633
Clavreul A, Etcheverry A, Tetaud C, Rousseau A, Avril T, Henry C, Mosser J, Menei P (2015) Identification of two glioblastoma-associated stromal cell subtypes with different carcinogenic properties in histologically normal surgical margins. J Neurooncol 122(1):1–10
Cucchiarini M, Ren XL, Perides G, Terwilliger EF (2003) Selective gene expression in brain microglia mediated via adeno-associated virus type 2 and type 5 vectors. Gene Ther 10(8):657–667
Rosario AM, Cruz PE, Ceballos-Diaz C, Strickland MR, Siemienski Z, Pardo M, Schob KL, Li A, Aslanidi GV, Srivastava A, Golde TE, Chakrabarty P (2016) Microglia-specific targeting by novel capsid-modified AAV6 vectors. Mol Ther Methods Clin Dev 3:16026
Gyorgy B, Maguire CA (2017) Extracellular vesicles: nature’s nanoparticles for improving gene transfer with adeno-associated virus vectors. Wiley Interdiscip Rev Nanomed Nanobiotechnol 10(3):e1488
Gyorgy B, Fitzpatrick Z, Crommentuijn MH, Mu D, Maguire CA (2014) Naturally enveloped AAV vectors for shielding neutralizing antibodies and robust gene delivery in vivo. Biomaterials 35(26):7598–7609
Hudry E, Martin C, Gandhi S, Gyorgy B, Scheffer DI, Mu D, Merkel SF, Mingozzi F, Fitzpatrick Z, Dimant H, Masek M, Ragan T, Tan S, Brisson AR, Ramirez SH, Hyman BT, Maguire CA (2016) Exosome-associated AAV vector as a robust and convenient neuroscience tool. Gene Ther 23(4):380
Mao P, Joshi K, Li J, Kim SH, Li P, Santana-Santos L, Luthra S, Chandran UR, Benos PV, Smith L, Wang M, Hu B, Cheng SY, Sobol RW, Nakano I (2013) Mesenchymal glioma stem cells are maintained by activated glycolytic metabolism involving aldehyde dehydrogenase 1A3. Proc Natl Acad Sci USA 110(21):8644–8649
Badr CE, Niers JM, Morse D, Koelen JA, Vandertop P, Noske D, Wurdinger T, Zalloua PA, Tannous BA (2011) Suicidal gene therapy in an NF-kappaB-controlled tumor environment as monitored by a secreted blood reporter. Gene Ther 18(5):445–451
Badr CE, Niers JM, Tjon-Kon-Fat LA, Noske DP, Wurdinger T, Tannous BA (2009) Real-time monitoring of nuclear factor kappaB activity in cultured cells and in animal models. Mol Imaging 8(5):278–290
Lee Y, Messing A, Su M, Brenner M (2008) GFAP promoter elements required for region-specific and astrocyte-specific expression. Glia 56(5):481–493
Maguire CA, Balaj L, Sivaraman S, Crommentuijn MH, Ericsson M, Mincheva-Nilsson L, Baranov V, Gianni D, Tannous BA, Sena-Esteves M, Breakefield XO, Skog J (2012) Microvesicle-associated AAV vector as a novel gene delivery system. Mol Ther 20(5):960–971
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Nagashima G, Suzuki R, Asai J, Fujimoto T (2002) Immunohistochemical analysis of reactive astrocytes around glioblastoma: an immunohistochemical study of postmortem glioblastoma cases. Clin Neurol Neurosurg 104(2):125–131
Lee J, Borboa AK, Baird A, Eliceiri BP (2011) Non-invasive quantification of brain tumor-induced astrogliosis. BMC Neurosci 12:9
Dashkoff J, Lerner EP, Truong N, Klickstein JA, Fan Z, Mu D, Maguire CA, Hyman BT, Hudry E (2016) Tailored transgene expression to specific cell types in the central nervous system after peripheral injection with AAV9. Mol Ther Methods Clin Dev 3:16081
Meliani A, Fitzpatrick F, Marmier S, Leborgne C, Collaud F, Sola MS, Charles S, Ronzitti G, Vignaud A, van Wittenberghe L, Marolleau B, Jouen F, Tan S, Boyer O, Christophe O, Brisson AR, Maguire CA, Mingozzi F (2017) Enhanced liver gene transfer and evasion of pre-existing humoral immunity with exosome-enveloped AAV vectors. Blood Adv 1(23):2019–2031
Gyorgy B, Sage C, Indzhykulian AA, Scheffer DI, Brisson AR, Tan S, Wu X, Volak A, Mu D, Tamvakologos PI, Li Y, Fitzpatrick Z, Ericsson M, Breakefield XO, Corey DP, Maguire CA (2017) Rescue of hearing by gene delivery to inner-ear hair cells using exosome-associated AAV. Mol Ther 25(2):379–391
Wassmer SJ, Carvalho LS, Gyorgy B, Vandenberghe LH, Maguire CA (2017) Exosome-associated AAV2 vector mediates robust gene delivery into the murine retina upon intravitreal injection. Sci Rep 7:45329
Bevan AK, Duque S, Foust KD, Morales PR, Braun L, Schmelzer L, Chan CM, McCrate M, Chicoine LG, Coley BD, Porensky PN, Kolb SJ, Mendell JR, Burghes AH, Kaspar BK (2011) Systemic gene delivery in large species for targeting spinal cord, brain, and peripheral tissues for pediatric disorders. Mol Ther 19(11):1971–1980
Bhat KP, Balasubramaniyan V, Vaillant B, Ezhilarasan R, Hummelink K, Hollingsworth F, Wani K, Heathcock L, James JD, Goodman LD, Conroy S, Long L, Lelic N, Wang S, Gumin J, Raj D, Kodama Y, Raghunathan A, Olar A, Joshi K et al (2013) Mesenchymal differentiation mediated by NF-kappaB promotes radiation resistance in glioblastoma. Cancer Cell 24(3):331–346
Biswas SK, Lewis CE (2010) NF-kappaB as a central regulator of macrophage function in tumors. J Leukoc Biol 88(5):877–884
Escarcega RO, Fuentes-Alexandro S, Garcia-Carrasco M, Gatica A, Zamora A (2007) The transcription factor nuclear factor-kappa B and cancer. Clin Oncol 19(2):154–161
Wilhelmsson U, Eliasson C, Bjerkvig R, Pekny M (2003) Loss of GFAP expression in high-grade astrocytomas does not contribute to tumor development or progression. Oncogene 22(22):3407–3411
Tada H, Maron DJ, Choi EA, Barsoum J, Lei H, Xie Q, Liu W, Ellis L, Moscioni AD, Tazelaar J, Fawell S, Qin X, Propert KJ, Davis A, Fraker DL, Wilson JM, Spitz FR (2001) Systemic IFN-beta gene therapy results in long-term survival in mice with established colorectal liver metastases. J Clin Invest 108(1):83–95
GuhaSarkar D, Neiswender J, Su Q, Gao G, Sena-Esteves M (2017) Intracranial AAV-IFN-beta gene therapy eliminates invasive xenograft glioblastoma and improves survival in orthotopic syngeneic murine model. Mol Oncol 11(2):180–193
Hottinger AF, Stupp R, Homicsko K (2014) Standards of care and novel approaches in the management of glioblastoma multiforme. Chin J Cancer 33(1):32–39
Shen D, Guo CC, Wang J, Qiu ZK, Sai K, Yang QY, Chen YS, Chen FR, Wang J, Panasci L, Chen ZP (2015) Interferon-alpha/beta enhances temozolomide activity against MGMT-positive glioma stem-like cells. Oncol Rep 34(5):2715–2721
Happold C, Roth P, Silginer M, Florea AM, Lamszus K, Frei K, Deenen R, Reifenberger G, Weller M (2014) Interferon-beta induces loss of spherogenicity and overcomes therapy resistance of glioblastoma stem cells. Mol Cancer Ther 13(4):948–961
Natsume A, Wakabayashi T, Ishii D, Maruta H, Fujii M, Shimato S, Ito M, Yoshida J (2008) A combination of IFN-beta and temozolomide in human glioma xenograft models: implication of p53-mediated MGMT downregulation. Cancer Chemother Pharmacol 61(4):653–659
Natsume A, Ishii D, Wakabayashi T, Tsuno T, Hatano H, Mizuno M, Yoshida J (2005) IFN-beta down-regulates the expression of DNA repair gene MGMT and sensitizes resistant glioma cells to temozolomide. Cancer Res 65(17):7573–7579
Park JH, Ryu CH, Kim MJ, Jeun SS (2015) Combination therapy for gliomas using temozolomide and interferon-beta secreting human bone marrow derived mesenchymal stem cells. J Korean Neurosurg Soc 57(5):323–328
Wakabayashi T, Kayama T, Nishikawa R, Takahashi H, Hashimoto N, Takahashi J, Aoki T, Sugiyama K, Ogura M, Natsume A, Yoshida J (2011) A multicenter phase I trial of combination therapy with interferon-beta and temozolomide for high-grade gliomas (INTEGRA study): the final report. J Neurooncol 104(2):573–577
Motomura K, Natsume A, Kishida Y, Higashi H, Kondo Y, Nakasu Y, Abe T, Namba H, Wakai K, Wakabayashi T (2011) Benefits of interferon-beta and temozolomide combination therapy for newly diagnosed primary glioblastoma with the unmethylated MGMT promoter: a multicenter study. Cancer 117(8):1721–1730
Brach MA, Hass R, Sherman ML, Gunji H, Weichselbaum R, Kufe D (1991) Ionizing radiation induces expression and binding activity of the nuclear factor kappa B. J Clin Invest 88(2):691–695
Veuger SJ, Hunter JE, Durkacz BW (2009) Ionizing radiation-induced NF-kappaB activation requires PARP-1 function to confer radioresistance. Oncogene 28(6):832–842
Sherman ML, Datta R, Hallahan DE, Weichselbaum RR, Kufe DW (1991) Regulation of tumor necrosis factor gene expression by ionizing radiation in human myeloid leukemia cells and peripheral blood monocytes. J Clin Invest 87(5):1794–1797
Iwamoto KS, McBride WH (1994) Production of 13-hydroxyoctadecadienoic acid and tumor necrosis factor-alpha by murine peritoneal macrophages in response to irradiation. Radiat Res 139(1):103–108
Rath BH, Fair JM, Jamal M, Camphausen K, Tofilon PJ (2013) Astrocytes enhance the invasion potential of glioblastoma stem-like cells. PLoS ONE 8(1):e54752
Acknowledgements
We thank the MGH Quantitative Real-Time PCR Core Facility for use of the quantitative PCR equipment for AAV quantitation and RT-qPCR analysis of transgene expression.
Funding
This work was supported by an American Brain Tumor Association Discovery Grant (CAM), a Cure Alzheimer’s Fund award (CAM) and the National Institutes of Health, the National Cancer Institute K22CA197053 (C.E.B.).
Author information
Authors and Affiliations
Contributions
CAM and CEB conceived of the study. AV, CAM, SGL, JSN, DJP, PSC, AM, ZF, EH, KP, SG, DM, DG, CEB performed experiments. CAM, CEB, SGL, AV, MSE, DG, ASR, BTH analyzed the results. CAM and CEB wrote the manuscript with input from all authors. All the authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
CAM has submitted patent applications regarding the exo-AAV platform. CAM holds equity in and is a founder and scientific advisor of Chameleon Biosciences, Inc, a gene therapy company.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Volak, A., LeRoy, S.G., Natasan, J.S. et al. Virus vector-mediated genetic modification of brain tumor stromal cells after intravenous delivery. J Neurooncol 139, 293–305 (2018). https://doi.org/10.1007/s11060-018-2889-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2889-2