Abstract
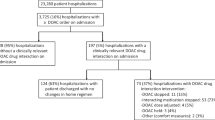
Data are limited on the effects of drug interactions on direct-acting oral anticoagulant (DOAC) levels. We evaluated the effects of the use of interacting drugs on DOAC levels in patients with atrial fibrillation (AF). We reviewed data of AF patients tested for DOAC levels in 2013–2017. The primary outcomes were drug levels exceeding the expected steady-state range, and in the highest quartile. A multivariate analysis was performed to evaluate the correlation of treatment by the use of interacting drugs, CYP3A4 and P-glycoprotein (P-gp) inhibitors, with the primary outcomes. Overall, 147 patients underwent DOAC level measurement [dabigatran (n = 31), rivaroxaban (n = 29), apixaban (n = 87)]. Thirty-three (22.4%) had drug levels exceeding the expected range. Seventy-nine (53.7%) patients were treated with at least one interacting drug. In multivariate analysis, the concomitant use of interacting drugs was an independent predictor for drug levels exceeding the expected range (OR 3.3, 95% CI 1.20–9.05). The defined daily dose of the interacting drug correlated positively with DOAC levels (r = 0.29, P = 0.001). Co-treatment with interacting drugs was associated with extremely high levels of dabigatran, (OR 16.6, 95% CI 1.29–215.18) but not of the other DOAC examined. Concomitant use of interacting drugs is associated with high DOAC levels in patients with AF. Further investigation is warranted to establish the differences between specific DOAC, evaluate the effect on patient outcomes, and characterize the role of DOAC monitoring in this setting.
Similar content being viewed by others
References
Douros A, Renoux C, Coulombe J et al (2017) Patterns of long-term use of non-vitamin K antagonist oral anticoagulants for non-valvular atrial fibrillation: Quebec observational study. Pharmacoepidemiol Drug Saf 26:1546–1554
Giustozzi M, Cristina M, Verdecchia P et al (2016) Vitamin K and non-vitamin K antagonist oral anticoagulants for non-valvular atrial fibrillation in real-life. Eur J Intern Med 33:42–46
Huisman MV, Rothman KJ, Paquette M et al (2017) The changing landscape for stroke prevention in AF. J Am Coll Cardiol 69:777–785. https://doi.org/10.1016/j.jacc.2016.11.061
Steffel J, Verhamme P, Potpara TS et al (2018) The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur Heart J 39:1330–1393
Wessler JD, Hil MP, Grip LT et al (2013) The P-glycoprotein transport system and cardiovascular drugs. J Am Coll Cardiol 61:2495–2502
Hellwig T, Gulseth M (2013) Pharmacokinetic and pharmacodynamic drug interactions with new oral anticoagulants: what do they mean for patients with atrial fibrillation ? Ann Pharmacother 47:1478–1487
Heidbuchel H, Verhamme P, Alings M et al (2013) European Heart Rhythm Association Practical Guide on the use of new oral anticoagulants in patients with non-valvular atrial fibrillation. Europace 15:625–651
Frost CE, Byon W, Song Y et al (2014) Effect of ketoconazole and diltiazem on the pharmacokinetics of apixaban, an oral direct factor Xa inhibitor. Br J Clin Pharmacol 79:838–846
Mueck W, Kubitza D, Becka M (2013) Co-administration of rivaroxaban with drugs that share its elimination pathways: pharmacokinetic effects in healthy subjects. Br J Clin Pharmacol 76:455–466
Katoh M, Nakajima M, Shimada N, Yamazaki H, Yokoi T (2000) Inhibition of human cytochrome P450 enzymes by 1,4-dihydropyridine calcium antagonists: prediction of in vivo drug-drug interactions. Eur J Clin Pharmacol 55:843–852
Nishio S, Watanabe H, Kosuge K et al (2005) Interaction between amlodipine and simvastatin in patients with hypercholesterolemia and hypertension. Hypertens Res 28:223–227
Katoh M, Nakajima M, Yamazaki H et al (2000) Inhibitory potencies of antagonists to P-glycoprotein-mediated transport: comparison with the effects on CYP3A4. Pharm Res 17:1–9
Drug Safety and Availability, FDA Drug Safety Communication, New restrictions, contraindications, and dose limitations for Zocor (simvastatin) to reduce the risk of muscle injury. https://www.fda.gov/Drugs/DrugSafety/ucm256581.htm
Drug Development and Drug Interactions (2018) Table of substrates, inhibitors and inducers, pp 1–14. https://www.fda.gov/Drugs/DevelopmentApprovalProcess/DevelopmentResources/DrugInteractionsLabeling/ucm093664.htm#table3-2
Eikelboom JW, Quinlan DJ, Hirsh J et al (2017) Laboratory monitoring of non-vitamin K antagonist oral anticoagulant use in patients with atrial fibrillation: a review. JAMA Cardiol 2:566–574
Dabigatran, Summary of product characteristics. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000829/WC500041059.pdf
Apixaban, highlights of prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202155s000lbl.pdf
Rivaroxaban, Prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/022406s000lbl.pdf
Dabigatran, Prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/022512s000lbl.pdf
Rottenstreich A, Zacks N, Kleinstern G et al (2018) Direct-acting oral anticoagulant drug level monitoring in clinical patient management. J Thromb Thrombolysis 45:543–549
Granger CB, Alexander JH, McMurray JJ et al (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365:981–992
Patel MR, Mahaffey KW, Garg J et al (2010) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:609–619
Connolly SJ, Ezekowitz MD, Yusuf S et al (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361:1139–1151
Giugliano RP, Ruff CT, Braunwald E et al (2013) Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med 369:2093–2104
Center for Drug Evaluation and Research Application Number, 202439Orig1s000, medical review, rivaroxaban. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2011/202439Orig1s000MedR.pdf
Center for Drug Evaluation and Research Application Number, 202155Orig1s000, medical review, apixaban. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/202155Orig1s000MedR.pdf
Center for Drug Evaluation and Research Application Number: 22-512, medical review, dabigatran. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022512Orig1s000MedR.pdf
Chang S-H, Chou I-J, Yeh Y-H et al (2017) Association between use of non-vitamin K oral anticoagulants with and without concurrent medications and risk of major bleeding in nonvalvular atrial fibrillation. JAMA 318:1250–1259
Ruff CT, Giugliano RP, Braunwald E et al (2015) Association between edoxaban dose, concentration, anti-factor Xa activity, and outcomes: an analysis of data from the randomised, double-blind ENGAGE AF-TIMI 48 trial. Lancet 6:2288–2295
Reilly PA, Lehr T, Haertter S et al (2014) The effect of dabigatran plasma concentrations and patient characteristics on the frequency of ischemic stroke and major bleeding in atrial fibrillation patients. J Am Coll Cardiol 63:321–328
Acknowledgements
We appreciate Ms. Cindy Cohen’s help in editing.
Author information
Authors and Affiliations
Contributions
BHR contributed to conception, design, acquisition, analysis, and interpretation. AR contributed to conception, design, acquisition and interpretation. NZ contributed to design and analysis. MM contributed to conception and interpretation. IM contributed to design and analysis. AP contributed to design, analysis, and interpretation. YK contributed to conception, design and interpretation. All authors critically revised manuscript and gave final approval.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflicts of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the local institutional review board of Hadassah Medical Center Helsinki Committee.
Informed consent
Informed consent was waived by the by the institutional review board of Hadassah Medical Center.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hirsh Raccah, B., Rottenstreich, A., Zacks, N. et al. Drug interaction as a predictor of direct oral anticoagulant drug levels in atrial fibrillation patients. J Thromb Thrombolysis 46, 521–527 (2018). https://doi.org/10.1007/s11239-018-1738-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-018-1738-7