Abstract
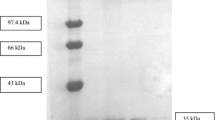
The genes of CS-2 lipase and its cognate foldase were cloned from Pseudomonas aeruginosa CS-2. A stop codon was not found in the lipase gene. The amino acid sequence deduced from the lipase gene from P. aeruginosa CS-2 showed 97.8%, 71.3%, and 71.2% identity with lipases from P. aeruginosa LST-03, P seudomonas mendocina ymp, and Pseudomonas stutzeri A1501, respectively. The co-expression of CS-2 lipase and its cognate foldase of P. aeruginosa CS-2 in E scherichia coli BL21 (DE3) resulted in the formation of a soluble lipase. The recombinant lipase and foldase were purified to homogeneity using nickel affinity chromatography and about 10.2-fold with 40.9% recovery was achieved for the purification of the recombinant lipase. The molecular masses of the lipase and the foldase were estimated to be 35.7 and 38.3 kDa in SDS-PAGE, respectively. The recombinant lipase showed stability in the presence of some organic solvents. The recombinant CS-2 lipase was immobilized and subsequently used for the synthesis of butyl acetate in heptane. The conversion of substrate decreased from 98.2% to 87.4% after 5 cycles in reuse of the immobilized lipase.





Similar content being viewed by others
References
Hasan, F., Shah, A. A., & Hameed, A. (2006). Enzyme and Microbial Technology, 39, 235–251.
Ghanem, A., & Aboul-Enein, H. Y. (2005). Chirality, 17, 1–15.
Houde, A., Kademi, A., & Leblanc, D. (2004). Applied Biochemistry and Biotechnology, 118, 155–170.
Theil, F. (1995). Chemical Reviews, 95, 2203–2227.
Ogino, H., Miyamoto, K., & Ishikawa, H. (1994). Applied and Environmental Microbiology, 60, 3884–3886.
Hun, C. J., Rahaman, R. N. Z. R. A., & Salleh, A. B. (2003). Biochemical Engineering Journal, 35, 147–157.
Rahaman, R. N. Z. R. A., Baharum, S. N., & Salleh, A. B. (2006). The Journal of Microbiology, 44, 583–590.
Zhao, L. L., Xu, J. H., Zhao, J., Pan, J., & Wang, Z. L. (2008). Process Biochemistry, 43, 626–633.
Ruchi, G., Anshu, G., & Khare, S. K. (2008). Process Biochemistry, 43, 1040–1046.
Dandavate, V., Jinjala, J., Keharia, H., & Madamwar, D. (2009). Bioresource Technology, 100, 3374–3381.
Ogino, H., Hiroshima, S., Hirose, S., Yasuda, M., Ishimi, K., & Ishikawa, H. (2004). Molecular Genetics and Genomics, 271, 189–196.
Ogino, H., Mimitsuka, T., Muto, T., Matsumura, M., Yasuda, M., Ishimi, K., et al. (2004). Journal of Molecular Microbiology and Biotechnology, 7, 212–223.
Ogino, H., Katou, Y., Akagi, R., Mimitsuka, T., Hiroshima, S., Gemba, Y., et al. (2007). Extremophiles, 11, 809–817.
Rahaman, R. N. Z. R. A., Chin, J. H., & Salleh, A. B. (2003). Molecular Genetics and Genomics, 269, 252–260.
Long, Z. D., Xu, J. H., Zhao, L. L., Pan, J., Yang, S., & Hua, L. (2007). Journal of Molecular Catalysis B Enzymatic, 47, 105–110.
Quyen, D. T., Schmidt-Dannert, C., & Schmid, R. D. (1999). Applied and Environmental Microbiology, 65, 787–794.
Rosenau, F., & Jaeger, K. E. (2000). Biochimie, 82, 1023–1032.
Zhang, A. J., Gao, R. J., Diao, N. B., Xie, G. Q., Gao, G., & Cao, S. G. (2009). Journal of Molecular Catalysis B Enzymatic, 56, 78–84.
Madan, B., & Mishra, P. (2010). Applied Microbiology and Biotechnology, 85, 597–604.
Park, Y. C., Shaffer, C. E., & Bennett, G. N. (2009). Applied Microbiology and Biotechnology, 85, 13–25.
Salah, R. B., Ghamghui, H., Miled, N., Mejdoub, H., & Youssef, G. (2007). Journal of Bioscience and Bioengineering, 103, 368–372.
Laemmli, U. K. (1970). Nature, 228, 680–685.
Bradford, M. M. (1976). Analytical Biochemistry, 72, 248–254.
Leitgeb, M., & Knez, Ž. (1990). Journal of the American Oil Chemists’ Society, 67, 775–778.
Peng, R., Lin, J., & Wei, D. (2010). Applied Biochemistry and Biotechnology, 162, 733–743.
Nagao, T., Shimada, Y., Sugihara, A., & Tominaga, Y. (1998). Journal of Biochemistry, 124, 1124–1129.
Kohno, M., Enatsu, M., Funatsu, J., Yoshiizumi, M., & Kugimiya, W. (2001). Journal of Biotechnology, 87, 203–210.
Yomo, T., Urabe, I., & Okada, H. (1992). Proceedings of the National Academy of Sciences of the United States of America, 89, 3780–3784.
Monnerjahn, C., Techel, D., Mohamed, S. A., & Rensing, L. A. (2000). FEMS Microbiology Letters, 183, 307–312.
Keiler, K. C., Shapiro, L., & Williams, K. P. (2002). Proceedings of the National Academy of Sciences of the United States of America, 97, 7778–7783.
Akrim, M., Bally, M., Ball, G., Tommassen, J., Teerink, H., Filloux, A., et al. (1993). Molecular Microbiology, 10, 431–443.
Luo, Y., Zheng, Y., Jiang, Z., Ma, Y., & Wei, D. (2006). Applied Microbiology and Biotechnology, 73, 349–355.
Traub, P. C., Schmidt-Dannert, C., Schmitt, J., & Schmid, R. D. (2001). Applied Microbiology and Biotechnology, 55, 198–204.
Verma, M. L., Azmi, W., & Kanwar, S. S. (2009). Acta Microbiologica et Immunologica Hungarica, 56, 229–242.
Acknowledgments
The financial support from the Ministry of Science and Technology of the People’s Republic of China is gratefully acknowledged (project IDs: 2006AA020203 and 2009CB724703).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Peng, R., Lin, J. & Wei, D. Co-Expression of an Organic Solvent-Tolerant Lipase and its Cognate Foldase of Pseudomonas aeruginosa CS-2 and the Application of the Immobilized Recombinant Lipase. Appl Biochem Biotechnol 165, 926–937 (2011). https://doi.org/10.1007/s12010-011-9309-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-011-9309-9