Abstract
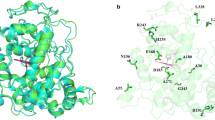
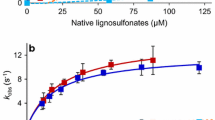
Expressed as insoluble forms in Escherichia coli, native cationic cell wall peroxidase (CWPO-C) from the poplar tree and mutant variants were successfully reactivated via refolding experiments and used to elucidate the previously presumed existence of an electron transfer (ET) pathway in the CWPO-C structure. Their catalytic properties were fully characterized through various analyses including steady-state kinetic, direct oxidation of lignin macromolecules and their respective stabilities during the polymerization reactions. The analysis results proved that the 74th residue on the CWPO-C surface plays an important role in catalyzing the macromolecules via supposed ET mechanism. By comparing the residual activities of wild-type CWPO-C and mutant 74W CWPO-C after 3 min, mutation of tyrosine 74 residue to tryptophan increased the radical resistance of peroxidase up to ten times dramatically while maintaining its capability to oxidize lignin macromolecules. Furthermore, extension of poly(catechin) as well as lignin macromolecules with CWPO-C Y74W mutant clearly showed that this radical-resistant peroxidase mutant can increase the molecular weight of various kinds of polyphenolics by using surface-located active site. The anti-oxidation activity of the synthesized poly(catechin) was confirmed by xanthine oxidase assay. The elucidation of a uniquely catalytic mechanism in CWPO-C may improve the applicability of the peroxidase/H2O2 catalyst to green polymer chemistry.







Similar content being viewed by others
References
Ronald, H., & Wilfred, V. (2001). Plant Physiol, 126, 1351–1357.
Takahama, U., & Oniki, T. (1997). Plant Cell Physiol, 38, 456–462.
Sahoo, S. K., Liu, W., Samuelson, L. A., Kumar, J., & Colli, A. L. (2002). Macromolecules, 35, 9990–9998.
Mejias, L., Reihmann, M. H., Sepulveda-Boza, S., & Ritter, H. (2002). Macromol Biosci, 2, 24–32.
Wariishi, H., Sheng, D., & Gold, M. H. (1994). Biochemistry, 33, 5545–5552.
Blodig, W., Doyle, W., Smith, A. T., & Piontek, K. (1998). Biochemistry, 37, 8832–8838.
Doyle, W. A., Blodig, W., Veitch, N. C., Piontek, K., & Smith, A. T. (1998). Biochemistry, 37, 15097–15105.
Sasaki, S., Nonaka, D., Wariishi, H., Tsutsumi, Y., & Kondo, R. (2008). Phytochemistry, 69, 348–355.
Shigeto, J., Itoh, Y., Tsutsumi, Y., & Kondo, R. (2012). FEBS Journal., 279, 348–357.
Kurisawa, M., Chung, J. E., Uyama, H., & Kobayyashi, S. (2003). Macromol Biosci, 3, 758–764.
Kurisawa, M., Chung, J. E., Kim, Y. J., Uyama, H., & Kobayashi, S. (2003). Biomacromolecules, 4, 469–471.
Higuchi, R., Krummel, B., & Saiki, R. (1988). Nucleic Acids Res, 16(15), 7351–7367.
Pham, L. T. M., Kim, S. J., Song, B. K., & Kim, Y. H. (2011). Protein Expr Purif, 80(2), 268–273.
Lopez-Serrano, M. A., & Ros, B. (1997). J Food Sci, 62(4), 676–723.
Weinkauf, R., Schanen, P., Yang, D., Soukara, S., & Schlag, E. W. (1995). J Phys Chem, 99, 11255–11265.
Weinkauf, R., Schanen, P., Metsala, A., Schlag, E. W., Burgle, M., & Kessler, H. (1996). J Phys Chem, 100, 18567–18585.
Lehr, L., Horneff, T., Weinkauf, R., & Schlag, E. W. (2005). J Phys Chem, 109, 8074–8080.
Morreel, K., Ralph, J., Kim, H., Lu, F., Goeminne, G., Ralph, S., Messens, E., & Boerjan, W. (2004). Plant Physiol, 136, 3537–3549.
Hewson, W. D., & Dunford, B. (1976). J Biol Chem, 251, 6036–6042.
Hewson, W. D., & Dunford, B. (1976). J Biol Chem, 251, 6043–6052.
Poulos, T. L. (1993). Curr Opin Biotechnol, 4, 484–489.
Arnao, M. B., Acosta, M., Del-Rio, J. A., & Garcia-Canovas, F. (1990). Biochim Biophys Acta, 1038, 85–89.
Valderrama, B., Ayala, M., & Vazuquez-Dehalt, R. (2002). Chem Biol, 9, 555–565.
Nakamoto, S., & Machida, N. (1992). Wat Res., 26, 49–54.
Wu, Y., Taylor, K. E., Biswas, N., & Bewtra, J. K. (1998). Enzyme Microb Technol, 22, 315–322.
Ghioureliotis, M., & Nicell, J. A. (1999). Enzyme Microb Technol, 25, 185–193.
Chang, H. C., Holland, R. D., & Bumpus, J. A. (1999). Chem Biol Interact, 123, 197–217.
Joback, K. G., & Reid, R. C. (1987). Chem Eng Commun., 57, 233–243.
Ferreira, D., Steynberg, J.P., Roux, D.G., Brandt, E.V., (1992) Tetrahedron, 48, 1743–1803.
Ferreira, D., & Li, X. C. (2000). Nat Prod Rep, 17, 193–212.
Acknowledgments
This work was supported by the Korean Government (NRF-2009-0093281, 2013K000361) and Kwangwoon University 2013; we, the authors, are thankful for this support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 2,255 kb).
Rights and permissions
About this article
Cite this article
Pham, L.T.M., Kim, S.J., Ahn, U.S. et al. Extension of Polyphenolics by CWPO-C Peroxidase Mutant Containing Radical-Robust Surface Active Site. Appl Biochem Biotechnol 172, 792–805 (2014). https://doi.org/10.1007/s12010-013-0534-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0534-2