Abstract
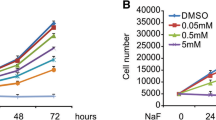
Advanced glycation end products (AGEs) accumulate under high-glucose conditions and affect the healing of bone damage through various pathways; however, the detail mechanisms underlying these changes are unknown. In this study, we investigated the effects of AGEs on the apoptosis of in vitro-cultured rat osteoblasts under high-glucose conditions and explored the underlying mechanisms of these effects. First, we cultured rat osteoblasts and determined the accumulation of AGEs in the culture medium under high-glucose conditions. Then, we cultured rat osteoblasts under a high glucose concentration (35 mM), a normal glucose concentration (5.5 mM), and a normal glucose concentration (5.5 mM) in the presence of AGEs. We examined the effects of high glucose and AGEs on the apoptosis of rat osteoblasts at different time points and further analyzed the activity and changes in the levels of procaspase-3, caspase-3, and the caspase-3 substrate poly ADP-ribose polymerase (PARP). Finally, we added sRAGE (soluble RAGE) (an AGE inhibitor) or DEVD (a caspase-3 inhibitor) to each culture group and examined apoptosis under each culture condition and the changes in the levels of procaspase-3, caspase-3, and its substrate PARP. The results showed that the high-glucose condition and the addition of AGEs increased the apoptosis of rat osteoblast cells and simultaneously increased the activity and quantity of caspase-3. These increases could be inhibited by the AGE inhibitor sRAGE or the caspase-3 inhibitor DEVD. The above results demonstrate that high-glucose conditions lead to the accumulation of AGEs and activation of the caspase-3 signaling pathway, resulting in the increased apoptosis of cultured rat osteoblast cells.






Similar content being viewed by others
References
Bass, E. F., Baile, C. A., Lewis, R. D., & Giraudo, S. Q. (2013). Bone quality and strength are greater in growing male rats fed fructose compared with glucose. Nutrition Research, 33(12), 1063–1071.
Ruzicska, E., & Poór, G. (2011). Diabetes and bone metabolism. Orvosi Hetilap, 152(29), 1156–1160.
Dhanasekaran, M., Indumathi, S., Rajkumar, J. S., & Sudarsanam, D. (2013). Effect of high glucose on extensive culturing of mesenchymal stem cells derived from subcutaneous fat, omentum fat and bone marrow. Cell Biochemistry and Function, 31(1), 20–29.
Okazaki, K., Yamaguchi, T., Tanaka, K., Notsu, M., Ogawa, N., Yano, S., & Sugimoto, T. (2012). Advanced glycation end products (AGEs), but not high glucose, inhibit the osteoblastic differentiation of mouse stromal ST2 cells through the suppression of osterix expression, and inhibit cell growth and increasing cell apoptosis. Calcified Tissue International, 91(4), 286–296.
Mercer, N., Ahmed, H., Etcheverry, S. B., Vasta, G. R., & Cortizo, A. M. (2007). Regulation of advanced glycation end product (AGE) receptors and apoptosis by AGEs in osteoblast-like cells. Molecular and Cellular Biochemistry, 306(1–2), 87–94.
Alikhani, M., Alikhani, Z., Boyd, C., MacLellan, C. M., Raptis, M., Liu, R., Pischon, N., Trackman, P. C., Gerstenfeld, L., & Graves, D. T. (2007). Advanced glycation end products stimulate osteoblast apoptosis via the MAP kinase and cytosolic apoptotic pathways. Bone, 40(2), 345–353.
Wu, B., Cui, J., Zhang, C., & Li, Z. (2012). A polysaccharide from Agaricus blazei inhibits proliferation and promotes apoptosis of osteosarcoma cells. International Journal of Biological Macromolecules, 50(4), 1116–1120.
Liu, Z., Jiang, H., Dong, K., Liu, S., Zhou, W., Zhang, J., Meng, L., Rausch-Fan, X., & Xu, X. (2015). Different concentrations of glucose regulate proliferation and osteogenic differentiation of osteoblasts via the PI3 kinase/Akt pathway. Implant Dentistry, 24(1), 83–91.
Guo, C., Zeng, X., Song, J., Zhang, M., Wang, H., Xu, X., Du, F., & Chen, B. (2012). A soluble receptor for advanced glycation end-products inhibits hypoxia/reoxygenation-induced apoptosis in rat cardiomyocytes via the mitochondrial pathway. International Journal of Molecular Sciences, 13(9), 11923–11940.
Kajikawa, M., Nakashima, A., Fujimura, N., Maruhashi, T., Iwamoto, Y., Iwamoto, A., Matsumoto, T., Oda, N., Hidaka, T., Kihara, Y., Chayama, K., Goto, C., Aibara, Y., Noma, K., Takeuchi, M., Matsui, T., Yamagishi, S., & Higashi, Y. (2015). Ratio of serum levels of AGEs to soluble form of RAGE is a predictor of endothelial function. Diabetes Care, 38(1), 119–125.
Wu, J. H., Yao, Y. L., Gu, T., Wang, Z. Y., Pu, X. Y., Sun, W. W., Zhang, X., Jiang, Y. B., & Wang, J. J. (2014). MiR-421 regulates apoptosis of BGC-823 gastric cancer cells by targeting caspase-3. Asian Pacific Journal of Cancer Prevention, 15(13), 5463–5468.
Gu, W., Zhang, Q., Yin, W., & Li, C. (2014). Caspase-3-mediated splenic lymphocyte apoptosis in a porcine model of cardiac arrest. American Journal of Emergency Medicine, 32(9), 1027–1032.
Szwed, M., Laroche-Clary, A., Robert, J., & Jozwiak, Z. (2014). Induction of apoptosis by doxorubicin-transferrin conjugate compared to free doxorubicin in the human leukemia cell lines. Chemico-Biological Interactions, 220, 140–148.
Węsierska-Gądek, J., & Heinzl, S. (2014). Interactions between ataxia telangiectasia mutated kinase inhibition, poly(ADP-ribose) polymerase-1 inhibition and BRCA1 status in breast cancer cells. J Cancer Prevention, 19(2), 125–136.
Ferrucci, A., Leboffe, L., Agamennone, M., Di Pizio, A., Fiocchetti, M., Marino, M., Ascenzi, P., & Luisi, G. (2015). Ac-tLeu-Asp-H is the minimal and highly effective human caspase-3 inhibitor: biological and in silico studies. Amino Acids, 47(1), 153–162.
Liu, J., Wang, Y., Yuan, X., Feng, Y., & Liu, H. (2010). Cyclic-stretch induces the apoptosis of myoblast by activation of caspase-3 protease in a magnitude-dependent manner. International Journal of Biochemistry and Cell Biology, 42(12), 2004–2011.
Satin, L. S., Butler, P. C., Ha, J., & Sherman, A. S. (2015). Pulsatile insulin secretion, impaired glucose tolerance and type 2 diabetes. Molecular Aspects of Medicine, 42, 61–77.
Irles, E., Ñeco, P., Lluesma, M., Villar-Pazos, S., Santos-Silva, J. C., Vettorazzi, J. F., Alonso-Magdalena, P., Carneiro, E. M., Boschero, A. C., Nadal, Á., & Quesada, I. (2015). Enhanced glucose-induced intracellular signaling promotes insulin hypersecretion: pancreatic beta-cell functional adaptations in a model of genetic obesity and prediabetes. Molecular and Cellular Endocrinology, 404, 46–55.
Wang, R., Liang, H., Li, H., Dou, H., Zhang, M., Baobuhe, Du, Z., Gao, M., & Wang, R. (2014). USF-1 inhibition protects against oxygen-and-glucose-deprivation-induced apoptosis via the downregulation of miR-132 in HepG2 cells. Biochemical and Biophysical Research Communications, 446(4), 1053–1059.
Zhang, X., Zhao, Y., Chu, Q., Wang, Z. Y., Li, H., & Chi, Z. H. (2014). Zinc modulates high glucose-induced apoptosis by suppressing oxidative stress in renal tubular epithelial cells. Biological Trace Element Research, 158(2), 259–267.
Acknowledgments
This work was supported by the Natural Science Foundation of China (No.81371178), Shanghai Municipal Education Commission Foundation (No. ZZjdyx12101),
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no competing interests. The manuscript has been read and approved by all authors.
Additional information
Jiaqiang Liu, Jing Mao, and Yi Jiang contributed equally to this work and should be considered as co-first author.
Rights and permissions
About this article
Cite this article
Liu, J., Mao, J., Jiang, Y. et al. AGEs Induce Apoptosis in Rat Osteoblast Cells by Activating the Caspase-3 Signaling Pathway Under a High-Glucose Environment In Vitro. Appl Biochem Biotechnol 178, 1015–1027 (2016). https://doi.org/10.1007/s12010-015-1925-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1925-3