Abstract
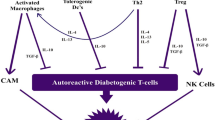
The distinction that in areas where helminthic infections are common, autoimmune diseases are less prevalent, led to the investigation of immune modulatory properties of helminths and their derivatives. Such are phosphorylcholine (PC) moieties which are a component of secreted products of helminths. PC has been broadly studied for its attenuating effects on the human immune system. In an attempt to develop a novel therapeutic small molecule for the treatment of autoimmune conditions, we have conjugated PC with tuftsin, a natural immunomodulatory tetrapeptide, to create TPC. Herein, we review our findings regarding the effects of TPC in murine models of three autoimmune diseases—systemic lupus erythematosus (SLE), inflammatory bowel disease (IBD), and rheumatic arthritis (RA), as well as ex-vivo samples from giant cell arteritis (GCA) patients. In all four disease models examined, TPC was shown to attenuate the inflammatory response by reducing expression of pro-inflammatory cytokines and altering the phenotype of T cell expression. In murine models, TPC has further produced a significant improvement in clinical disease scores with no significant side effects noted. Our findings suggest TPC presents promising potential as a novel therapeutic agent for the effective treatment of various autoimmune conditions.
Similar content being viewed by others
Abbreviations
- TPC:
-
Tuftsin-phosphorylcholine
- GCA :
-
Giant cell arteritis
- IBD :
-
Internal bowel disease
- PBMCs:
-
Periferal blood monouclear cells
- RA:
-
Rheumatoid arthritis
- SLE:
-
Systemic lupus erythematosus
References
Hotez PJ, Brindley PJ, Bethony JM, King CH, Pearce EJ, Jacobson J. Helminth infections: the great neglected tropical diseases. J Clin Invest. 2008 Apr 1;118(4):1311–21.
Greenwood BM. Autoimmune disease and parasitic infections in Nigerians. Lancet. 1968;292(7564):380–2.
Rook GAW, Raison CL, Lowry CA. Microbial ‘old friends’, immunoregulation and socioeconomic status. Clin Exp Immunol. 2014;177(1):1–12.
Strachan DP. Hay fever, hygiene, and household size. BMJ. 1989;299(6710):1259–60.
Versini M, Jeandel P-Y, Bashi T, Bizzaro G, Blank M, Shoenfeld Y. Unraveling the hygiene hypothesis of helminthes and autoimmunity: origins, pathophysiology, and clinical applications. BMC Med. 2015;13:81.
Summers RW, Elliott DE, Urban JF, Thompson R, Weinstock JV. Trichuris suis therapy in Crohn’s disease. Gut. 2005;54(1):87–90.
Summers RW, Elliott DE, Urban JF, Thompson RA, Weinstock JV. Trichuris suis therapy for active ulcerative colitis: a randomized controlled trial. Gastroenterology. 2005;128(4):825–32.
Zaccone P, Cooke A. Helminth mediated modulation of type 1 diabetes (T1D). Int J Parasitol. 2013;43(3–4):311–8.
Fleming J, Hernandez G, Hartman L, Maksimovic J, Nace S, Lawler B, et al. Safety and efficacy of helminth treatment in relapsing-remitting multiple sclerosis: results of the HINT 2 clinical trial. Mult Scler Houndmills Basingstoke Engl. 2017;1352458517736377:1.
Benzel F, Erdur H, Kohler S, Frentsch M, Thiel A, Harms L, et al. Immune monitoring of Trichuris suis egg therapy in multiple sclerosis patients. J Helminthol. 2012;86(3):339–47.
Sewell D, Qing Z, Reinke E, Elliot D, Weinstock J, Sandor M, et al. Immunomodulation of experimental autoimmune encephalomyelitis by helminth ova immunization. Int Immunol. 2003;15(1):59–69.
Liu Q, Sundar K, Mishra PK, Mousavi G, Liu Z, Gaydo A, et al. Helminth infection can reduce insulitis and type 1 diabetes through CD25- and IL-10-independent mechanisms. Infect Immun. 2009;77(12):5347–58.
Selmi C. Are helminths to be trusted as allies in the war against autoimmunity and chronic inflammation? Isr Med Assoc J IMAJ. 2016;18(3–4):139–40.
McInnes IB, Leung BP, Harnett M, Gracie JA, Liew FY, Harnett W. A novel therapeutic approach targeting articular inflammation using the filarial nematode-derived phosphorylcholine-containing glycoprotein ES-62. J Immunol. 2003;171(4):2127–33.
Pineda MA, Lumb F, Harnett MM, Harnett W. ES-62, a therapeutic anti-inflammatory agent evolved by the filarial nematode Acanthocheilonema viteae. Mol Biochem Parasitol. 2014;194(1):1–8.
Goodridge HS, McGUINESS S, Houston KM, Egan CA, Al-Riyami L, Alcocer MJC, et al. Phosphorylcholine mimics the effects of ES-62 on macrophages and dendritic cells. Parasite Immunol. 2006;29(3):127–37.
Siebert A, Gensicka-Kowalewska M, Cholewiński G, Tuftsin DK. Properties and analogs. Curr Med Chem. 2017;24:3711–27.
Najjar VA, Nishioka K. ‘Tuftsin’: a natural phagocytosis stimulating peptide. Nature. 1970 Nov;228(5272):672–3.
Siemion IZ, Kluczyk A. Tuftsin: on the 30-year anniversary of Victor Najjar’s discovery. Peptides. 1999;20(5):645–74.
Bashi T, Blank M, Ben-Ami Shor D, Fridkin M, Versini M, Gendelman O, et al. Successful modulation of murine lupus nephritis with tuftsin-phosphorylcholine. J Autoimmun. 2015;59:1–7.
Shemer A, Kivity S, Shovman O, Perry O, Ben-Ami Shor D, Bragazzi NL, et al. Tuftsin-phosphorylcholine (TPC) equally effective to methylprednisolone in ameliorating lupus nephritis in a mice model. Clin Exp Immunol 2018;In Press, 193, 160, 166.
Bashi T, Shovman O, Fridkin M, Volkov A, Barshack I, Blank M, et al. Novel therapeutic compound tuftsin-phosphorylcholine attenuates collagen-induced arthritis. Clin Exp Immunol. 2016;184(1):19–28.
Ben-Amram H, Bashi T, Werbner N, Neuman H, Fridkin M, Blank M, et al. Tuftsin-Phosphorylcholine maintains normal gut microbiota in collagen induced arthritic mice. Front Microbiol. 2017;8:1222.
Croci S, Bonacini M, Muratore M, Caruso A, Fontana A, Boiardi L, et al. First evidences of immunomodulatory activities of tuftsin-phosphorylcholine on samples from patients with giant cell arteritis in comparison to corticosteroids. 2018; Submitted.
Ben-Ami Shor D, Harel M, Eliakim R, Shoenfeld Y. The hygiene theory harnessing helminths and their ova to treat autoimmunity. Clin Rev Allergy Immunol. 2013;45(2):211–6.
Bashi T, Bizzaro G, Ben-Ami Shor D, Blank M, Shoenfeld Y. The mechanisms behind helminth’s immunomodulation in autoimmunity. Autoimmun Rev. 2015;14(2):98–104.
Anne C. Review series on helminths, immune modulation and the hygiene hypothesis: how might infection modulate the onset of type 1 diabetes? Immunology. 2008;126(1):12–7.
Wu Z, Nagano I, Asano K, Takahashi Y. Infection of non-encapsulated species of Trichinella ameliorates experimental autoimmune encephalomyelitis involving suppression of Th17 and Th1 response. Parasitol Res. 2010;107(5):1173–88.
Ruyssers NE, Winter D, Y B, Man D, G J, Loukas A, et al. Therapeutic potential of helminth soluble proteins in TNBS-induced colitis in mice. Inflamm Bowel Dis. 2009;15(4):491–500.
Correale J, Farez M. Association between parasite infection and immune responses in multiple sclerosis. Ann Neurol. 2007;61(2):97–108.
Osada Y, Shimizu S, Kumagai T, Yamada S, Kanazawa T. Schistosoma mansoni infection reduces severity of collagen-induced arthritis via down-regulation of pro-inflammatory mediators. Int J Parasitol. 2009;39(4):457–64.
Maślińska M, de Luca F, Sharif K. Tuftsin-phosphorylcholine treatment of autoimmune diseases – a benefit and a message from helminths? Reumatologia. 2017;55(6):267–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Segal, Y., Blank, M. & Shoenfeld, Y. Tuftsin phosphorylcholine—a novel compound harnessing helminths to fight autoimmunity. Immunol Res 66, 637–641 (2018). https://doi.org/10.1007/s12026-018-9051-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-018-9051-2