Abstract
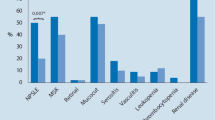
Metabolic parameters like uric acid, lipids and homocysteine are influenced by immunopathological mechanisms underlying the autoimmune disease processes. The current study examined the differences in these parameters and the correlation between inflammatory and metabolic variables in rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) patients. The cross-sectional prospective study included 24 treatment-naïve patients with moderate to severe diseases-15 subjects had RA and 9 had SLE. Atherogenic index of plasma (AIP) was used to assess the cardiovascular risk of the patients. Spearman’s correlation was performed to verify the relationship between inflammatory and metabolic parameters. A two-tailed P < 0.05 was considered statistically significant for all the analysis. SLE patients had higher uric acid levels, very low density lipoprotein-cholesterol, total cholesterol/high density lipoprotein-cholesterol ratio (TC/HDL-C) and logarithmic ratio of triglycerides to HDL-cholesterol (log[TG/HDL-C]) than RA. Whereas, reduced total lymphocyte count, lipoprotein(a), and low density lipoprotein cholesterol were noted in the former than latter group. Majority of the SLE patients had increased risk of cardiovascular diseases (> 0.24 AIP score) and RA patients in comparison had lower risk. Correlation among serum uric acid, lipid profile constituents and AIP was noted. The immunological process of SLE has greater impact on the metabolic parameters. Higher uric acid levels are suggestive of dysfunctional lipid profile. Understanding the implications of risk factors and its inflammatory role in autoimmune processes may assist in disease management.
Similar content being viewed by others
References
Charles-Schoeman C, Wang X, Lee YY, Shahbazian A, Navarro-Millán I, Yang S, et al. Association of triple therapy with improvement in cholesterol profiles over two year follow-up in the TEAR Trial. Arthritis Rheumatol. 2016;68(3):577–86.
Wang H, Ye J. Regulation of energy balance by inflammation: common theme in physiology and pathology. Rev Endocr Metab Disord. 2015;16(1):47–54.
Shoenfeld Y, Gerli R, Doria A, Matsuura E, Cerinic MM, Ronda N, et al. Accelerated atherosclerosis in autoimmune rheumatic diseases. Circulation. 2005;112(21):3337–47.
McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365(23):2205–19.
Mok CC, Lau CS. Pathogenesis of systemic lupus erythematosus. J Clin Pathol. 2003;56(7):481–90.
Baechler EC, Batliwalla FM, Karypis G, Gaffney PM, Ortmann WA, Espe KJ, et al. Interferon-inducible gene expression signature in peripheral blood cells of patients with severe lupus. PNAS. 2003;100(5):2610–5.
Chung CP, Oeser A, Solus JF, Gebretsadik T, Shintani A, Avalos I, et al. Inflammation-associated insulin resistance: differential effects in rheumatoid arthritis and systemic lupus erythematosus define potential mechanisms. Arthritis Rheum. 2008;58(7):2105–12.
Kahlenberg JM, Kaplan MJ. Mechanisms of premature atherosclerosis in rheumatoid arthritis and lupus. Annu Rev Med. 2013;64(1):249–63.
Ward MM. Premature morbidity from cardiovascular and cerebrovascular diseases in women with systemic lupus erythematosus. Arthritis Rheum. 1999;42(2):338–46.
Solomon DH, Karlson EW, Rimm EB, Cannuscio CC, Mandl LA, Manson JE, et al. Cardiovascular morbidity and mortality in women diagnosed with rheumatoid arthritis. Circulation. 2003;107(9):1303–7.
Ostojic P, Bartolovic D. Disease activity, obesity, functional disability, and depression in patients with rheumatoid arthritis: impact on lipid status, glycoregulation, and risk for coronary heart disease. Z Rheumatol. 2016;75(7):716–22.
Kono H, Chen C-J, Ontiveros F, Rock KL. Uric acid promotes an acute inflammatory response to sterile cell death in mice. J Clin Invest. 2010;120(6):1939–49.
Meek IL, Vonkeman HE, van de Laar MA. Hyperuricaemia: a marker of increased cardiovascular risk in rheumatic patients: analysis of the ACT-CVD cohort. BMC Musculoskelet Disord. 2014;15:174.
Huang C, Li M, Liu Y, Wang Q, Guo X, Zhao J, et al. Baseline characteristics and risk factors of pulmonary arterial hypertension in systemic lupus erythematosus patients. Medicine (Baltimore). 2016;95(10):e2761.
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62(9):2569–81.
Petri M, Orbai A-M, Alarcón GS, Gordon C, Merrill JT, Fortin PR, et al. Derivation and validation of systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64(8):2677–86.
Madsen OR. Is DAS28-CRP with three and four variables interchangeable in individual patients selected for biological treatment in daily clinical practice? Clin Rheumatol. 2011;30(12):1577–82.
Bhardwaj S, Bhattacharjee J, Bhatnagar MK, Tyagi S. Atherogenic index of plasma, castelli risk index and atherogenic coefficient-new parameters in assessing cardiovascular risk. Int J Pharm Biol Sci. 2013;3(3):359–64.
Adu EM, Ukwamedu HA, Oghagbon ES. Assessment of cardiovascular risk indices in type 2 diabetes mellitus. Trop Med Surg. 2015;3:184.
Ranjit PM, Guntuku G, Pothineni R. Comparison of lipid profile and new atherogenic indices among the coronary artery disease (CAD)-negative and -positive diabetic dyslipidemia subjects. Int J Med Sci Public Health. 2015;4(11):1574–9.
Dobiásová M. AIP–atherogenic index of plasma as a significant predictor of cardiovascular risk: from research to practice. Vnitr Lek. 2006;52(1):64–71.
Cohen J. Statistical power analysis for the behavioral sciences. Hillsdale, NJ: L. Erlbaum Associates; 1988.
Santos MJ, Vinagre F, Silva JJ, Gil V, Fonseca JE. Cardiovascular risk profile in systemic lupus erythematosus and rheumatoid arthritis: a comparative study of female patients. Acta Reumatol Port. 2010;35(3):325–32.
Gaál K, Tarr T, Lőrincz H, Borbás V, Seres I, Harangi M, et al. High-density lipopoprotein antioxidant capacity, subpopulation distribution and paraoxonase-1 activity in patients with systemic lupus erythematosus. Lipids Health Dis. 2016;15:60.
Batún GJA, Olán F, Hernández NÉ. Dyslipidemia and atherogenic risk in patients with rheumatoid arthritis. Clin Investig Arterioscler. 2016;28(3):123–31.
Liao KP, Playford MP, Frits M, Coblyn JS, Iannaccone C, Weinblatt ME, et al. The association between reduction in inflammation and changes in lipoprotein levels and HDL cholesterol efflux capacity in rheumatoid arthritis. J Am Heart Assoc. 2015;4(2):e001588.
Toms TE, Panoulas VF, Kitas GD. Dyslipidaemia in rheumatological autoimmune diseases. Open Cardiovasc Med J. 2011;24(5):64–75.
Kim S-H, Lee C-K, Lee EY, Park SY, Cho YS, Yoo B, et al. Serum oxidized low-density lipoproteins in rheumatoid arthritis. Rheumatol Int. 2004;24(4):230–3.
Liao KP, Cai T, Gainer VS, Cagan A, Murphy SN, Liu C, et al. Lipid and lipoprotein levels and trend in rheumatoid arthritis compared to the general population. Arthritis Rheum. 2013;65(12):2046–50.
Al Gadban MM, Alwan MM, Smith KJ, Hammad SM. Accelerated vascular disease in systemic lupus erythematosus: role of macrophage. Clin Immunol. 2015;157(2):133–44.
Zegkos T, Kitas G, Dimitroulas T. Cardiovascular risk in rheumatoid arthritis: assessment, management and next steps. Ther Adv Musculoskelet Dis. 2016;8(3):86–101.
Haeryfar SMM, Rassaian N, Moslemizadeh M, Hoseini-Gohari L. Elevated lipoprotein(a) in systemic lupus erythematosus. Arch Iran Med. 2002;5(4):230–4.
Missala I, Kassner U, Steinhagen-Thiessen E. A systematic literature review of the association of lipoprotein(a) and autoimmune diseases and atherosclerosis. Int J Rheumatol. 2012;2012:e480784.
Delgado Alves J, Ames PRJ, Donohue S, Stanyer L, Nourooz-Zadeh J, Ravirajan C, et al. Antibodies to high-density lipoprotein and beta2-glycoprotein I are inversely correlated with paraoxonase activity in systemic lupus erythematosus and primary antiphospholipid syndrome. Arthritis Rheum. 2002;46(10):2686–94.
Kiss E, Seres I, Tarr T, Kocsis Z, Szegedi G, Paragh G. Reduced paraoxonase1 activity is a risk for atherosclerosis in patients with systemic lupus erythematosus. Ann N Y Acad Sci. 2007;1108(1):83–91.
Romero FI, Khamashta MA, Hughes GR. Lipoprotein(a) oxidation and autoantibodies: a new path in atherothrombosis. Lupus. 2000;9(3):206–9.
Govindan KPS, Basha S, Ramesh V, Kumar CN, Swathi S. A comparative study on serum lipoprotein(a) and lipid profile between rheumatoid arthritis patients and normal subjects. J Pharm Bioallied Sci. 2015;7(Suppl 1):S22–5.
Kim K-J, Baek I-W, Park Y-J, Yoon C-H, Kim W-U, Cho C-S. High levels of uric acid in systemic lupus erythematosus is associated with pulmonary hypertension. Int J Rheum Dis. 2015;18(5):524–32.
Castillo-Martínez D, Marroquín-Fabián E, Lozada-Navarro AC, Mora-Ramírez M, Juárez M, Sánchez-Muñoz F, et al. Levels of uric acid may predict the future development of pulmonary hypertension in systemic lupus erythematosus: a seven-year follow-up study. Lupus. 2016;25(1):61–6.
Castejon R, Jimenez-Ortiz C, Rosado S, Tutor-Ureta P, Mellor-Pita S, Yebra-Bango M. Metabolic syndrome is associated with decreased circulating endothelial progenitor cells and increased arterial stiffness in systemic lupus erythematosus. Lupus. 2016;25(2):129–36.
Sheikh M, Movassaghi S, Khaledi M, Moghaddassi M. Hyperuricemia in systemic lupus erythematosus: is it associated with the neuropsychiatric manifestations of the disease? Rev Bras Reumatol Engl Ed. 2016;56(6):471–7.
Amador-Patarroyo MJ, Rodriguez-Rodriguez A, Montoya-Ortiz G. How does age at onset influence the outcome of autoimmune diseases? Autoimmune Dis. 2012;2012:251730.
Scalzi LV, Hollenbeak CS, Wang L. Racial disparities for age at time of cardiovascular events and cardiovascular death in SLE patients. Arthritis Rheum. 2010;62(9):2767–75.
Fransen J, Kazemi-Bajestani SMR, Bredie SJH, Popa CD. Rheumatoid arthritis disadvantages younger patients for cardiovascular diseases: a meta-analysis. PLoS ONE. 2016;11(6):e0157360.
McMahon M, Hahn BH, Skaggs BJ. Systemic lupus erythematosus and cardiovascular disease: prediction and potential for therapeutic intervention. Expert Rev Clin Immunol. 2011;7(2):227–41.
Acknowledgements
We acknowledge the help of www.research-assist.com for editing the manuscript. We thank the ChanRe Diagnostic laboratory, Bengaluru, for blood sample analysis and providing details of reports. The study was approved by the hospital Institutional Ethics Committee (IEC-CRICR/077/14).
Funding
This work was supported by the Immunology and Arthritis Research and Education trust (IARET), Bangalore, an independent trust supporting research in arthritis and education.
Author information
Authors and Affiliations
Contributions
Chandrashekara S conceptualized and monitored the study, Sachin Vithalrao Dhote patient recruitment and data cleaning, and Anupama K R conducted the statistical analysis and interpretation of the data. All the authors had access to anonymous data. All authors contributed for preparation of the manuscript and approved the finalized manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Chandrashekara, S., Dhote, S.V. & Anupama, K.R. The Differential Influence of Immunological Process of Autoimmune Disease on Lipid Metabolism: A Study on RA and SLE. Ind J Clin Biochem 34, 52–59 (2019). https://doi.org/10.1007/s12291-017-0715-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-017-0715-9