Abstract
Purpose: Oral etoposide has been shown to be effective against some solid tumor types. This phase II study examined the efficacy of oral etoposide in patients with untreated advanced adenocarcinoma of the pancreas.
Methods and patients: Previously untreated patients with measurable unresectable or metastatic disease were eligible for inclusion in this study. They were required to have a Zubrod performance status of ≤ 2 and normal hematological, renal, and hepatic functions. Written informed consent was obtained from all patients. The etoposide dose was 50 mg/m2/d given orally daily for 21 days with a 7-day rest period and was adjusted based on toxicity. Response was assessed after two courses of therapy.
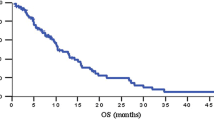
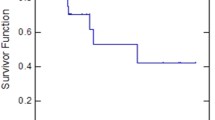
Results: Eighteen patients were enrolled and fourteen were evaluable for toxicity and response. Twelve patients developed progressive disease while two patients had only a minor response. The median number of courses was 2 (range, 1–4) and there were 29 total courses delivered. The median survival was 3.5 months (range, 1.6–25.9). There were no treatment-related deaths. Toxicity was moderate.
Conclusions: Oral etoposide is not effective against advanced adenocarcinoma of the pancreas.
Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA: Cancer statistics, 1998. Cancer J Clin 48: 6–29, 1998
Brower S, Hoff P, Jones D, Cunningham J, Myerson R: Pancreatic cancer, hepatobiliary cancer, and neuroendocrine cancers of the GI tract. In: Pazdur R, Coia L, Hoskins W, Wagman L (eds) Cancer Management: A Multidisciplinary Approach. PRR, NY, 1998, pp 113–148
Schnall S, Macdonald J: Chemotherapy of adenocarcinoma of the pancreas. Semin Oncol 23: 220–228, 1996
Burris HA, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P, Nelson R, Dorr FA, Stephens CD, Von Hoff DD: Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15: 2403–2413, 1997
DeJong RS, Mulder NH, Dijksterhuis D, DeVries EGE: Review of current clinical experience with prolonged (oral) etoposide in cancer treatment. Anticancer Res 15: 2319–2330, 1995
Ajani J, Dumas P, Mansfield P: Phase II trial of oral etoposide in untreated patients with advanced gastric carcinoma. Cancer J Sci Am (in press, 1999)
Estape J, Palombo H, Grau JJ, Daniels M, Vinolas N, Mane JM: Phase II study of chronic oral etoposide in solid tumors. Proc Am Soc Clin Oncol 11: 355, 1992
Sternberg CN, Magill GB, Cheng EW, Applewhite A, Sordillo PP: Etoposide (VP-16) in the treatment of advanced adenocarcinoma of the pancreas. Am J Clin Oncol 11: 172–173, 1988
Asbury RF, Cnaan A, Johnson L, Harris J, Zaentz SD, Haller DG: An Eastern Cooperative Oncology Group phase II study of single agent DHAD, VP-16, aclacinomycin, or spirogermanium in metastatic pancreatic cancer. Am J Clin Oncol 17: 166–170, 1994
Hainsworth J, Johnson D, Frazier S, Greco F: Chronic daily administration of oral etoposide-a phase I trial. J Clin Oncol 7: 396–401, 1989
Ajani JA, Welch SR, Raber MN, Fields WS, Krakoff IH: Comprehensive criteria for assessing therapy-induced toxicity. Cancer Invest 8: 147–159, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thomas, E., Dumas, P. & Ajani, J.A. Oral etoposide for patients with advanced adenocarcinoma of the pancreas. Invest New Drugs 16, 333–335 (1998). https://doi.org/10.1023/A:1006113421167
Issue Date:
DOI: https://doi.org/10.1023/A:1006113421167