Abstract
Purpose : To compare the efficacy, efficacy and safety of a fixed daily dose of recombinant FSH (Puregon®) of a 100- and 200-IU regimen in Asian women undergoing ovarian stimulation for IVF/ICSI.
Methods : This was a prospective, randomized, double-blind, multicenter (n = 9) study. Prior to the start of rFSH, all women were pretreated with a gonadotropin releasing hormone agonist (GnRH-a) for pituitary downregulation.
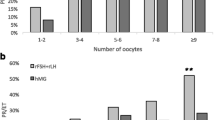
Results : A total of 330 women were treated with rFSH: 163 subjects with 100 IU and 167 subjects with . In the 200 IU treatment group, significantly more oocytes were retrieved compared to the 100 IU group (9.6 vs. 5.0 oocytes, p < 0.001). The total dose rFSH needed to develop at least three follicles with a diameter of ≥17 mm was significantly lower in the 100 IU treatment group (1194 vs. 2034 IU, p < 0.001). Although more cycle cancellations were seen in the 100 IU group (24 vs. 13%), the ongoing pregnancy rate per started cycle was comparable between both groups (16.6% in the 100 IU group vs. 15.0% in the 200 IU group).
Conclusions : The use of a 100 IU fixed dose is less effective in terms of the number of oocytes retrieved and the higher cancellation rate, but more efficient as indicated by a lower total recombinant FSH dose needed.
Similar content being viewed by others
REFERENCES
Out HJ, Mannaerts BMJL, Driessen SG, Coelingh Bennink HJ: A prospective, randomized, assessor-blind, multicenter study comparing recombinant follicle-stimulating hormone (Puregon vs Metrodin) in in-vitro fertilization. Hum Reprod 1995;10:2534-2540
Devroey P, Tournaye H, van Steirteghem A, Hendrix P, Out HJ: The use of a 100 IU starting dose of recombinant follicle stimulating hormone (Puregon) in in-vitro fertilization. Hum Reprod 1998;13:565-566
Out HJ, Lindenberg S, Mikkelsen AL, Eldar-Geva T, Healy DL, Leader A, Rodriguez-Escudero FJ, Garcia-Velasco JA, Pellicer A: A prospective, randomized, double-blind clinical trial to study the efficacy and efficiency of a fixed dose of recombinant follicle stimulating hormone (Puregon) in women undergoing ovarian stimulation. Hum Reprod 1999;14:622-627
Out HJ, David I, Ron-El R, Friedler S, Shalev E, Geslevich J, Dor J, Shulman A, Ben-Rafael Z, Fisch B, Dirnfeld M: A randomized, double-blind clinical trial using fixed daily doses of 100 or 200 IU of recombinant FSH in ICSI cycles. Hum Reprod 2001;16:1104-1109
CPMP Working Party on Efficacy of Medicinal Products: Good Clinical Practice for trials on medicinal products in the European Community. Pharmacol Toxicol 1990;67: 361-372
Cochran WG: The combination of estimates from different experiments. Biometrics 1954;10:101-129
Cochran WG: Some methods for strengthening the common tests. Biometrics 1954;10:417-451
Land JA, Yarmolinskaya MI, Dumoulin JCM, Evers JLH: High-dose human menopausal gonadotropin stimulation in poor responders does not improve in vitro fertilization outcome. Fertil Steril 1996;65:961-965
van Hooff MHA, Alberda AT, Huisman GJ, Zeilmaker GH, Leerentveld RA: Doubling the human menopausal gonadotrophin dose in the course of an in-vitro fertilization treatment cycle in low responders: A randomized study. Hum Reprod 1993;8:369-373
Jones HW Jr, Out HJ, Hoomans EHM, Driessen SGAJ, Coelingh Bennink HJT: Cryopreservation: the practicalities of evaluation. Hum Reprod 1997;12:1522-1524
Out HJ, Driessen SGAJ, Mannaerts BMJL, Coelingh Bennink HJT: Recombinant follicle stimulating hormone (follitropin beta, Puregon) yields higher pregnancy rates in in vitro fertilization than urinary gonadotropins. Fertil Steril 1997;68:138-142
Ubaldi F, Camus M, Smitz J, Coelingh Bennink H, van Steirteghem A, Devroey P: Premature luteinization in in vitro fertilization cycles using gonadotropin-releasing hormone agonist (GnRH-a) and recombinant follicle-stimulating hormone (FSH) and GnRH-a and urinary FSH. Fertil Steril 1996;66:275-280
Out HJ, Braat DDM, Lintsen BME, Gurgan T, Bukulmez O, Gokmen O, Keles G, Caballero P, Gonzalez JM, Fabregues F, Balasch J, Roulier R: Increasing the daily dose of recombinant follicle stimulating hormone Puregon does not compensate for the age-related decline in retrievable oocytes after ovarian stimulation. Hum Reprod 2000;15:29-35
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asian Puregon Study Group, Hoomans, E.H.M. & Mulder, B.B. A Group-Comparative, Randomized, Double-Blind Comparison of the Efficacy and Efficiency of Two Fixed Daily Dose Regimens (100- and 200-IU) of Recombinant Follicle Stimulating Hormone (rFSH, Puregon®) in Asian Women Undergoing Ovarian Stimulation for IVF/ICSI. J Assist Reprod Genet 19, 470–476 (2002). https://doi.org/10.1023/A:1020358419073
Issue Date:
DOI: https://doi.org/10.1023/A:1020358419073